Abstract
The latest advances in asthma treatment have highlighted the significance of eosinophilia and the possible role of some pro-eosinophilic mediators, like interleukins (IL) IL-5, IL-4/IL-13, and IL-33 in the disease’s pathogenesis. Considering that a subgroup of patients with chronic obstructive pulmonary disease (COPD) may have blood eosinophilia akin to that seen in asthma, numerous studies in the last decade have suggested that eosinophilic COPD is a separate entity. While the exact role of blood eosinophils in the pathophysiology of COPD remains unclear, eosinophilia seems to increase the effectiveness of corticosteroid therapy. Currently, monoclonal antibodies targeting the interleukins (IL-5, IL-4, IL-13, and IL-33) or their receptors are being investigated in patients with COPD belonging in T2-high endotype. This review focuses on the mechanisms of eosinophilia in COPD, the effects of eosinophilia on disease outcome, and examines the most recent data on the use of peripheral blood eosinophilia in treating patients with COPD. Finally, we emphasize the current implication of monoclonal antibodies in COPD in the context of eosinophilic airway inflammation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Eosinophilic chronic obstructive pulmonary disease (COPD) has recently been shown to be a distinct disease. | |
The effectiveness of inhaled corticosteroids (ICS) increases as eosinophil counts rise in stable COPD. | |
Blood eosinophil levels can be used as a predictor of response to systemic corticosteroids in COPD exacerbations. | |
Currently, monoclonal antibodies directed against interleukins (ILs) or their receptors (IL-5, IL-4, IL-13, and IL-33) are being tested in the T2 endotype of COPD. |
Introduction
Chronic obstructive pulmonary disease (COPD) is one of the most common diseases and a leading cause of respiratory failure, disability, and mortality worldwide. It is characterized by progressive and partially irreversible bronchial obstruction due to persistent structural changes mostly affecting distal airways and lung parenchyma [1, 2]. Airway obstruction is caused by chronic inflammation, which leads to numerous conditions, including peribronchial fibrosis, inflammatory infiltration and edema of the bronchial mucosa, bronchial smooth muscle spasm, loss of lung elasticity, destruction of the lung parenchyma and the presence of secretions in the bronchial lumen [3]. According to current knowledge, COPD is mainly a Th1-mediated inflammatory condition characterized by neutrophil preponderance and an increase in alveolar macrophages and CD8+ T cells, while asthma, which is a different chronic airway inflammatory disease, is often characterized by eosinophilic inflammation, which involves Th2-mediated airway immune response and allergic sensitization [4, 5]. Given that one-third of patients with COPD have a history of asthma, and over 40% of patients with airflow limitation have asthma–COPD overlap (ACO) [6], it has been suggested that asthma and COPD might be two different manifestations of the same disease [7].
Although, for many years, eosinophilic inflammation has been related to asthma and has been used for the differential diagnosis between asthma and COPD [8], recent evidence has shown that eosinophilic inflammation is also observed in 19–67% of patients with COPD [9]. This suggests that asthma and COPD sometimes share the same T2 endotype of inflammation [10]. Blood eosinophil (EOS) count is a widely used indicator of eosinophilic airway inflammation that can be used to predict the prognosis and severity of asthma [11]. However, there is currently no EOS threshold for the diagnosis of COPD or asthma, and the usefulness of blood eosinophils in predicting the probability of an exacerbation and providing treatment guidance for COPD and asthma is still debatable [12]. The number of eosinophils in sputum during a COPD exacerbation and the amount of eosinophils in blood or sputum during stable COPD can both be helpful indicators of how the patient would react to inhaled corticosteroids (ICS) and whether systemic corticosteroids should be used during COPD exacerbations [13]. Therefore, this review briefly summarizes the eosinophilic phenotype of COPD and the research progress of blood and sputum eosinophil levels as a biomarker in COPD.
In undertaking this review, we conducted a search of literature in PubMed, up to August 2024. Key search terms used were “COPD”, “eosinophils”, “corticosteroid”, “exacerbations”, and “monoclonal antibodies”. The authors evaluated both original research papers and relevant reviews, which were shortlisted accordingly. Only papers written in English were used. Every effort was made not to omit any significant study in the field, although we focused mainly on recent advances. This article is based on previously conducted studies and contains no new studies with human participants or animals performed by any of the authors.
Mechanisms of Eosinophilia
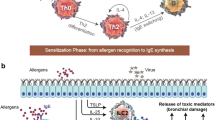
Eosinophils are produced in the bone marrow from CD34+ progenitor cells and their differentiation is induced by interleukin (IL)-5 and granulocyte–macrophage colony-stimulating factor [14]. Accumulation of eosinophils into the respiratory tract is promoted by T-cells producing IL-5 and other specific chemo-attractants [15] while locally generated IL-4 and 13 increase eosinophil adhesion to endothelial cells [16]. Several cytokines, chemokines, growth factors, and specific basic proteins found in the acidophilic cytoplasmic granules of eosinophils are extremely toxic to bronchial epithelial cells [17]. These products participate in disease manifestation and progression by inhibiting airway cilia function, stimulating bronchoconstriction, and promoting airway remodeling and immune response (Fig. 1). Eosinophils are not seen in the airways of healthy individuals and thus, their presence suggests the development of a pathological inflammatory response [18].
Eosinophils are involved in immune control, tissue damage and repair, elimination of hazardous compounds, the release of active substances, tumor cell growth inhibition and promotion of apoptosis [19]. Their primary function in allergic diseases is host immunological defense and post-parasitic immunity. A link has been seen between the number of EOS and the rate at which viruses and harmful bacteria are cleared, indicating that EOS may have antibacterial capacities [14]. Blood eosinophil levels are affected by several factors including allergic diseases, parasitic infections, dermatosis, autoimmune diseases, myeloproliferative malignancies, hypereosinophilic diseases such as eosinophilic granulomatosis with polyangiitis (EGPA), eosinophilic gastroenteritis, and drugs [i.e., antibiotics (penicillin, cephalosporin) and/or non-steroidal anti-inflammatory (aspirin, ibuprofen)] [14, 20].
Eosinophilia in COPD
The literature’s definition of peripheral blood eosinophilia is imprecise, but it has grown more specific as anti-cytokine treatments for eosinophilic diseases have evolved. Until recently, an absolute eosinophil count of 500–999 cells/μl was considered mild eosinophilia, 1000–1499 cells/μl was considered moderate eosinophilia, and > 1500 cells/μl was considered severe eosinophilia [21]. Under normal circumstances, the percentage of eosinophils in the blood ranges between 1 and 6%. Several experts have recently suggested that eosinophil counts 351–500 cells/μl should be classified as mild eosinophilia. Clinical trials on COPD have used a variety of cutoff points for “increased blood eosinophils”, such as eosinophil count exceeding 150 total cells/μl or more than 2% of total cells, even though these numbers fall within the normal range for eosinophil count. For example, in a post hoc analysis of 3177 patients with COPD and a history of exacerbations, 66% had an eosinophil count of 2% or higher at study entry [13], whereas in another study involving > 1100 individuals with COPD and a history of exacerbations, the median eosinophil count was reported to be 181 cells/μl with the upper quartile having a median eosinophil count of 280 eosinophils/μl [22]. In contrast with the current knowledge from research in asthma, wherein patients with eosinophilia have mostly been clearly demarcated using remarkably higher counts, the definition of “increased blood eosinophils” in patients with COPD is within acknowledged standard ranges [23]. The rationale behind this may lie in the pragmatic considerations involved in conducting clinical studies on medications that affect eosinophil counts. In other words, more patients may be included into a clinical trial if the definition of eosinophilia is lowered, and eventually, if benefit is exhibited, a greater expansion of the underlying market may exist for a drug whose preliminary trials used a lower inclusion value. Another explanation may be the fact that EOS count may vary notably in the same person during the day [24]. Additionally, the number of blood eosinophils can be reduced during a bacterial infection or after the administration of inhaled or systemic glucocorticosteroids [25,26,27,28]. Notably, compared to those with higher eosinophil counts, patients with COPD with lower blood eosinophil counts have less variability [29].
Additionally, several studies have demonstrated high blood and/or sputum eosinophil counts in a significant percentage of patients with COPD [4, 30, 31]. Cluster analysis of sputum granulocyte profiles has proposed eosinophil-rich sputum eosinophilia as a sign of a distinct eosinophil COPD phenotype [32], although the counts of eosinophils in airways do not seem to correlate strongly with disease progression as showed by GOLD staging [33]. However, SPIROMICS has shown that patients with elevated sputum eosinophil counts have worse lung function and more pronounced emphysema than those with low sputum eosinophil counts [34]. Additionally, compared to the low eosinophil group, the high eosinophil count group experienced more episodes of acute exacerbations of COPD requiring corticosteroid treatment. Furthermore, Siva et al. [35] showed that among stable COPD patients, a COPD therapy approach aimed to reduce eosinophilic airway inflammation was linked to a decrease in acute exacerbations of COPD of these patients.
On the other hand, lung tissue-infiltrating eosinophils in COPD remain largely unexplored. Previous studies have confirmed eosinophil presence in central and distal compartments [36, 37]. However, important questions remain about the anatomical localization and infiltration patterns and the theoretical underpinning for eosinophilia in COPD and its immunological triggers. Recently, it has been identified that a spatially confined eosinophil-rich type 2 microenvironment might represent a novel type of heterogeneity in the immunopathology of COPD that is likely to have implications for personalized treatment [38].
Finally, as clinicians, we should not forget that 30% of patients with COPD and 26% of patients with asthma have symptoms associated with asthma-chronic obstructive pulmonary disease (COPD) overlap syndrome (ACO) [39, 40]. A systematic review and meta-analysis [41] concluded that the percentage of induced sputum neutrophils are higher and levels of FEV1% predicted, diffusion lung capacity for carbon monoxide (DLCO%) predicted and fractional exhaled nitric oxide (FeNO) were lower in ACO patients than in pure asthmatic patients, however, the opposite was true when compared to patients with COPD alone. In addition, ACO patients have higher levels of serum total IgE, blood eosinophil counts and percentage of induced sputum eosinophils than those with pure COPD.
Eosinophilia as an Indicator of COPD Outcomes
Several studies have demonstrated that blood eosinophilia, as a single variable, is associated with worse outcomes in patients with COPD. In an observational cohort study including 479 patients admitted to hospital due to an exacerbation of COPD, 36% of participants met the definition of eosinophilia, which was defined as > 200 cells/μl or > 2% total cells [42]. This group of patients had a higher likelihood of readmission due to COPD and a higher number of visits to the emergency department within a year following their initial admission. A prospective multicenter observational study conducted in Greece further supported these findings. More specifically, 388 patients hospitalized for COPD exacerbations were divided into three groups (< 50, 50–149, and ≥ 150 cells/μl), according to peripheral blood eosinophils [43]. The study showed that higher blood eosinophils counts were related with better outcomes in these patients. Furthermore, a post hoc analysis of this study aiming to determine the efficiency of EOS for anticipating the requirement of noninvasive ventilation (NIV) in patients experiencing a COPD exacerbation revealed that low EOS levels during admission were associated with more severe disease and may indicate the need of NIV use [44]. On the contrary, a previous study including 243 hospitalized patients with COPD and eosinophilia (defined as blood eosinophils ≥ 200 cells/μl and/or ≥ 2% of the total leukocyte count) [45], concluded that the requirement length of stay was significantly shorter compared to patients without eosinophilia, while no difference in the number of readmissions in the following year was observed.
Additionally, according to a Danish cohort study including 2600 patients with COPD who were followed up for a median time of 3.3 years, a higher eosinophil count (> 343 cells/μl) was associated with a higher likelihood of exacerbations [31]. In a different study including patients with COPD exacerbations, those who did not have increased blood eosinophils had a lower mortality rate, shorter duration of hospital stay, and a negligible increase in the likelihood of readmission [46]. However, no difference in the rate of exacerbations was detected in Korean long-term outpatients in whom eosinophilia was defined as > 300 cells/μl, although patients with increased blood eosinophils showed increased survival throughout a 6-year observation period [47].
Until now, conflicting data exists regarding the exacerbation rates in patients with COPD and the predictive usefulness of blood eosinophil counts, with studies showing either no [48] or a positive relationship [49]. Nevertheless, the usage of inhaled corticosteroids and different past exacerbation history seem to be confounding factors in their assessment [27]. A study of 2400 patients with COPD (smokers and ex-smokers) showed no association between blood eosinophilia (> 200 cells/μl) and risk of exacerbations, although a weak correlation was found between sputum eosinophilia and COPD outcomes [34]. Also, in 458 patients with COPD, current or ex-smokers, in France, no correlation was seen between the exacerbation rate and eosinophils above the 2% threshold in comparison to those without eosinophilia [2]. The authors concluded that the association of higher blood eosinophils counts with exacerbations seen in previous studies may be population specific, which does not support generalizing the use of these cells as a possible biomarker for phenotyping of COPD. Recently, Price et al. [50], using evidence extracted from the Optimum Patient Care Research Database, concluded that blood eosinophil counts did not add significantly to the prediction of future exacerbation risk. Table 1 summarizes most important studies showing the association between blood eosinophil levels and outcomes in patients with COPD.
These findings stand in stark contrast to other studies including patients with adult asthma where a considerable increase in eosinophilia is related to significantly worse outcomes, such as increased frequency and severity of exacerbations [51, 52]. In fact, patients with eosinophilic asthma who are clinically resistant to corticosteroids seem to have worse outcomes [21]. Consequently, if eosinophilia in COPD can be used to identify a subgroup of patients, the link between eosinophilia and corticosteroid responsiveness may provide crucial information.
Eosinophils as a Biomarker of Inhaled Corticosteroid Responsiveness in Patients with COPD
Until now, there is no evidence linking eosinophilia to corticosteroid resistance in COPD; in fact, the results of several studies show the exact opposite. There is strong data showing that in patients with COPD with a history of exacerbations, the addition of ICS significantly reduced exacerbation rate [13]. More particularly, patients with blood eosinophil counts ≥ 2%, reduced exacerbations by 29% compared to those with blood eosinophil counts < 2% in whom it was not possible to show that ICS had a statistically meaningful benefit. In addition, individuals with significantly elevated counts of eosinophils show a progressively greater benefit from ICS [13]. In an analysis of the INSPIRE study who compared a specific ICS/LABA (inhaled corticosteroid/long-acting beta-agonist) combination to tiotropium, patients with COPD with ≥ 2% eosinophilia during study entry had a remarkable reduction in exacerbations (p = 0.006), but not the < 2% group (p = 0.186) [53]. Similar were the results from the analysis of the TRISTAN trial, which compared the combination of ICS and LABA with a placebo [53]. After adding ICS, there was a substantial reduction in exacerbations for the group with ≥ 2% eosinophils (p < 0.001), but not for the group with < 2% eosinophilia (p = 0.957).
Data, however, do not always support the hypothesis that eosinophilia determines which subgroup of patients with COPD is most likely to benefit with inhaled corticosteroids. In the ISOLDE study, eosinophil counts > 200 cells/μl were related to improved lung function in patients receiving ICS; however, the effect on exacerbation rate was inconsistent, and there was no difference in the eosinophilic group’s time to the first moderate or severe exacerbation [54]. Patients with eosinophil counts > 200 cells/μl under treatment with ICS, exhibited a lower annual rate of moderate to severe exacerbations. Furthermore, in the IMPACT study, which included 10,333 patients with COPD, single triple therapy (ICS/LABA/LAMA) (inhaled corticosteroids/long-acting beta-agonists/long-acting muscarinic antagonists) was compared with ICS/LABA or LABA/LAMA (long-acting beta-agonists/long-acting muscarinic antagonists) combinations; in 43% of the participants, the blood eosinophil count was lower than 150 cells/μl [55]. Regardless of eosinophil count, the rate of moderate/severe exacerbations was lower with triple therapy than with a LABA/LAMA combination. A post hoc analysis of the WISDOM study showed that patients with high blood EOS levels experienced more COPD exacerbations after ICS discontinuation. The authors proposed a threshold of > 4%, or > 300 cells/μl, to identify patients in whom ICS discontinuation might lead to COPD exacerbations [56]. Finally, in the FLAME trial (comparing LABA/LAMA combination to ICS/LABA), LABA/LAMA combination was more effective in the exacerbation reduction compared to the ICS/LABA in all blood eosinophil levels although in patients with higher numbers of blood eosinophils the two different therapies had similar effects [57].
Several meta-analyses have been conducted and different thresholds of blood eosinophil levels have been used to assess the role of ICS in patients with COPD, due to contradictory evidence provided in individual studies and post hoc analyses of clinical trials. These analyses have shown that ICS are considerably more effective in patients with higher eosinophil levels and do not play a significant role in reducing exacerbations in those with blood eosinophil count lower than 150 cells/μl [58, 59]. According to the GOLD guidelines, blood eosinophil count greater than 300 cells/μl, when combined with additional variables such as exacerbation rate, history of hospitalization for exacerbation, or history of asthma, should be considered strongly supportive of treating the patient with ICS, even though a true cutoff has not been defined [60]. In addition, all current evidence shows that a history infrequent exacerbations together with eosinophil counts < 150 cells/μl should be taken into consideration as negative predictors of reduction in exacerbation rate with ICS. Indeed, post-hoc analyses from the KRONOS study, who included patients with moderate to very severe COPD, propose an escalation to triple therapy in those with persistent/worsening symptoms and blood eosinophils > 100 cells/mm3, even if disease severity is moderate and there is no history of exacerbations [61]. Moreover, while blood eosinophils count can fluctuate from patient to patient, lower eosinophils values are often less variable, which makes them a fairly accurate indicator of the lack of response to ICS, especially in patients who have not previously experienced exacerbations [29]. This partially explains why a post hoc analysis of the FLAME study [62], indicates preferentially using blood eosinophil count off ICS or blood eosinophil count change during ICS treatment for guiding treatment decisions.
Currently, there is insufficient data to establish a correlation between eosinophilia and pneumonia risk in ICS users. The incidence of pneumonia in patients with blood eosinophils > 2% versus those with lower levels was evaluated in a recent trial of 10 studies utilizing ICS for COPD [63]. A marginally detected higher risk of pneumonia in patients on ICS with low eosinophils did not reach statistical significance in any research, and the pooled data did not demonstrate a meaningful difference in pneumonia risk while on ICS. Very recently, a post hoc analysis of the IMPACT study, also advocated that blood eosinophil count did not have an impact on the pneumonia risk in patients receiving triple therapy [64].
Eosinophils as a Biomarker of Systemic Corticosteroids Responsiveness in COPD Exacerbations
In the last decade there is strong evidence showing that patients with COPD experiencing a disease exacerbation respond more rapidly and effectively to systemic corticosteroids when their blood eosinophil count is ≥ 2% compared to lower levels of blood eosinophils [45, 65, 66]. More specifically, a case control trial showed that these patients with high eosinophil count when treated with OCS needed a shorter hospital stay [66]. Similar were the results of another study regarding the duration of hospital stay which found that patients with blood eosinophil levels ≥ 200 cells/μl who received prednisolone had shorter length of hospital stay compared to those who experienced exacerbations that were not eosinophilic [45]. Bafadhel et al., in a randomized, biomarker-directed, double-blind study, further investigated the relationship between EOS and oral corticosteroids [65]. Patients in the ‘standard of care’ arm received oral prednisolone to manage exacerbations of COPD, and these in the ‘biomarker-directed’ arm received prednisolone or placebo according to blood eosinophil count (> 2 versus ≤ 2% blood eosinophils, respectively). Prednisolone was not administered to 49% of patients in the biomarker-directed group. The Chronic Respiratory Questionnaire scores were similarly improved in the two groups, and these findings result in the conclusion that a reduction in the number of inappropriate OCS prescriptions is possible. Additionally, using available data from three randomized, controlled trials with 243 patients, the same investigators retrospectively applied groupings of ≥ 2% versus < 2% blood eosinophils and presence/absence of prednisolone therapy [67]. This meta-analysis concluded that patients experiencing a COPD exacerbation who have EOS ≥ 2% had significantly decreased treatment failure rates with prednisolone, compared with placebo, whereas on the other hand, prednisolone does not appear to improve outcomes in patients with a blood eosinophil count < 2%. The authors came to the conclusion that a biomarker-directed corticosteroid therapy strategy that maximizes benefit and minimizes worthless systemic corticosteroid administration may be a viable method based on the blood eosinophil count evaluated at the initiation of an exacerbation [67]. Finally, the CORTICO-COP study investigated if the use of systemic corticosteroid in patients hospitalized due to acute exacerbation of COPD could be safely reduced on the day of admission and every morning for 5 days using an algorithm based on blood eosinophil counts [68]. A total of 318 patients were randomized into two groups: the first group received systemic corticosteroids based on daily blood eosinophil counts (37.5 mg prednisolone on days when eosinophils were ≥ 300 cells/μl, and treatment was not administered at a level of < 300 cells/μl). The second group received standard treatment (37.5 mg prednisolone daily for 4 days). On the first day, all patients received 80 mg of methylprednisolone intravenously. In terms of the number of days alive and out of the hospital, eosinophil-guided therapy was not inferior to conventional care; however, the median duration of systemic corticosteroid therapy was shorter in the eosinophil-guided group (2 days) than in the control group (5 days).
Monoclonal Antibodies in the Treatment of Eosinophilic COPD
Initial trials of anti-IL-5 treatments showed that they were successful in significantly reducing the number of eosinophils in the sputum and blood of patients with asthma [69, 70]. Indeed the degree of eosinophilia is highly correlated with the effectiveness of anti-IL-5 therapy [71, 72]. Mepolizumab, benralizumab, and reslizumab are the three anti-IL-5 drugs that are now licensed for the treatment of bronchial asthma, and patients with COPD are currently enrolled in clinical trials for the first two of them (mepolizumab and benralizumab).
Following four weeks of treatment, mepolizumab (a humanized monoclonal IgG1 antibody that binds directly to IL-5), causes a 78% decrease in blood eosinophils and a 50–55% decrease in eosinophils in the bone marrow and lung tissue. Patients with eosinophil levels > 150 cells/μl at the beginning of treatment or > 300 cells/μl throughout the previous 12 months who have severe eosinophilic asthma are eligible for receiving mepolizumab. It is evident that a significant proportion of patients with COPD also exhibit this degree of eosinophilia [73].
Benralizumab is a humanized IgG1 antibody that targets the alpha chain of the IL-5 receptor. It can both block the effects of IL-5 and, through cellular cytotoxicity processes, cause tissue eosinophil depletion [72]. Benralizumab is recommended for individuals with severe eosinophilic asthma who have eosinophil levels > 300 cells/μl over a 6-week treatment period, as it depletes over 90% of tissue and blood eosinophils in 4 weeks.
The first extensive trials on mepolizumab for COPD were positive [74]. Two phase III, randomized, placebo-controlled, double-blind, parallel-group trials comparing mepolizumab (100 mg in METREX, 100 or 300 mg in METREO) with placebo, aimed to evaluate the annual rate of moderate or severe exacerbations, in patients with COPD with a history of exacerbations. In METREX study which included 462 patients with an eosinophilic phenotype (> 150 cells/μl at baseline or > 300 cells/μl in the last year), the annual rate of exacerbations was 1.40 per year in the mepolizumab group and 1.71 per year in the placebo group (p = 0.04). On the other hand, in METREO study which included 674 patients, the mean rate of exacerbations per year did not differ significantly between the 100 or 300 mg mepolizumab or placebo groups (1.19, 1.27, and 1.49 per year, respectively). However, patients with higher blood eosinophil counts at screening had a stronger effect of mepolizumab compared with placebo on the annual risk of moderate or severe exacerbations, and mepolizumab’s safety profile was comparable to that of placebo.
Brightling et al. was one of the first investigators who aimed to determine if benralizumab reduces the risk of COPD exacerbations in patients with high blood eosinophil levels [75]. In a randomized, double-blind, placebo-controlled, phase 2a study, 101 patients, randomly assigned to receive placebo or 100 mg benralizumab. Unfortunately, benralizumab did not lower the rate of acute COPD exacerbations as compared to placebo. GALATHEA and TERRANOVA were two trials which included 2665 patients with COPD in whom, over a period of 56 weeks, benralizumab was added to dual or triple inhaled therapy [76]. The study concluded that in patients with an elevated baseline blood eosinophil level, who received triple therapy and experienced ≥ 3 exacerbations in the previous year, benralizumab 100 mg every 8 weeks was more effective compared with placebo in terms of reducing exacerbations. However, there was no statistically significant reduction in moderate/severe exacerbations [76]. Results from the ABRA study on the efficacy of benralizumab monotherapy or its combination with prednisolone in patients with exacerbation of eosinophilic COPD are expected soon [77].
Finally, a Cochrane database analysis of six studies involving 5542 patients with COPD randomly assigned to anti-IL-5 medication showed that mepolizumab likely decreased the rate of moderate-to-severe exacerbations in patients with COPD with more than 150 eosinophils [78]. Furthermore, benralizumab decreased the hospitalization rate in patients whose eosinophil count was greater than 220 cells/μl [78]. Although individual studies of anti-IL-5 therapy failed to demonstrate a clear benefit, the promise in pooled data may indicate that, with appropriate patient selection, anti-IL-5 therapy may be beneficial for a highly specific subgroup of patients with COPD, or that the number needed to treat COPD with IL-5 treatment is so high that individual studies do not have the power to identify a difference.
Interleukin-13 plays a crucial role in the pathophysiology of asthma, through induction of goblet cell hyperplasia contraction of smooth muscle and airway hyper-responsiveness [79], and together with IL-4, activates the secretion of immunoglobulin E (IgE) and production of C–C cytokines that attract eosinophils to the site of inflammation. It is important to know that the receptors for interleukins 4 and 13 have the same alpha chain, and as a result an antibody targeting the alpha chain of both cytokines would affect both. Until today, only two antibodies have been tested against IL-13, tralokinumab and lebrikizumab. Although tralokinumab has not yet been assessed in COPD, lebrikizumab is currently being studied in eosinophilic COPD (NCT02546700). However, dupilumab, a fully human anti-interleukin-4 receptor α monoclonal antibody, which inhibits interleukin-4 and interleukin-13 signaling, has been resulted in significant increase in lung function and in reduction in severe exacerbations in patients with uncontrolled persistent asthma irrespective of baseline eosinophil count [80]. The same were the results when 300 mg dupilumab were administered in patients with COPD who, despite of receiving the standard triple therapy, had a blood eosinophil count of at least 300 per microliter and a high exacerbation risk, in a phase 3, double-blind, randomized trial [81]. Compared to those who received placebo, patients receiving dupilumab experienced fewer exacerbations, improved pulmonary function and quality of life, and had less severe respiratory symptoms. Recently, the NOTUS study also showed that dupilumab was related to improved lung function and reduced COPD exacerbations compared to placebo in patients with COPD and type 2 inflammation [82]. Finally, the COURSE study, a phase IIa multicenter, randomized, double-blind, placebo-controlled, parallel group trial designed to evaluate the safety and efficacy of tezepelumab (anti-thymic stromal lymphopoietin mAb) in patients with moderate to very severe COPD receiving triple therapy, and having had two or more documented COPD exacerbations in the 12 months, is still ongoing (NCT04039113).
Itepekimab, another monoclonal antibody acting against IL-33, which in turn induce Th2 type responses, activates mast cells, and stimulates the secretion of eosinophil chemotactic proteins and production of IL-13 [83]. In 2021, Rabe KF. and co-workers published a double-blind, phase 2a trial, which compared itepekimab with placebo in current or former smokers with moderate-to-severe COPD despite standard therapy [84]. The primary endpoint was not met, no reduction in exacerbation rate was observed, nor an improvement in lung function as evaluated by change in pre-bronchodilator FEV1 (forced expiratory volume in 1st second). Nevertheless, a subgroup analysis showed that itepekimab reduced exacerbation rate and improved lung function in former smokers with COPD, indicating that the underlying role of concurrent TH2 responses in the pathophysiology of COPD may be obscured by active smoking, leading to a preponderance of neutrophilic inflammation. Table 2 summarizes all the above studies referring to the possible role of monoclonal antibodies in the treatment of eosinophilic COPD.
Conclusions
Blood eosinophils play a significant role in COPD, and although their pathogenetic involvement has not yet been fully understood, numerous studies have shown that eosinophil counts can be utilized to guide treatment decisions. There is data showing a potential role for blood eosinophils in predicting COPD exacerbations, while the effectiveness of ICS seems to increase as eosinophil levels increase. In the absence of any evidence indicating that patients with COPD are more susceptible to the effects of eosinophils, the generally recognized definitions of eosinophilia used in most trials—> 2% of total cells or > 150 cells/μl—seem arbitrary and have dubious validity because they fall within the acknowledged normal range. Large, prospective studies that divide patients into several groups based on blood eosinophil counts are necessary to elucidate the function of eosinophils in predicting the effectiveness of ICS in COPD.
Biological treatments targeting cytokines or cytokine receptors implicated in eosinophilic inflammation, significantly improve asthma, but still have a restricted, although promising therapeutic effects in COPD. Prospective well-designed trials will help to understand how eosinophils contribute to the pathophysiology of COPD, how to choose the most targeted treatments for a given patient and assess the effectiveness of biological therapy in patients with COPD.
Data Availability
All data used are available in the references provided.
References
Ferrera MC, Labaki WW, Han MK. Advances in chronic obstructive pulmonary disease. Annu Rev Med. 2021;72:119–34.
Zysman M, Deslee G, Caillaud D, Chanez P, Escamilla R, Court-Fortune I, et al. Relationship between blood eosinophils, clinical characteristics, and mortality in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2017;12:1819–24.
Kim V, Rogers TJ, Criner GJ. New concepts in the pathobiology of chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2008;5:478–85.
George L, Brightling CE. Eosinophilic airway inflammation: role in asthma and chronic obstructive pulmonary disease. Ther Adv Chronic Dis. 2016;7:34–51.
Albertson TE, Chenoweth JA, Pearson SJ, Murin S. The pharmacological management of asthma-chronic obstructive pulmonary disease overlap syndrome (ACOS). Expert Opin Pharmacother. 2020;21:213–31.
Fouka E, Papaioannou AI, Hillas G, Steiropoulos P. Asthma–COPD overlap syndrome: recent insights and unanswered questions. J Pers Med. 2022. https://doi.org/10.3390/jpm12050708.
Leung C, Sin DD. Asthma–COPD overlap: what are the important questions? Chest. 2022;161:330–44.
Cukic V, Lovre V, Dragisic D, Ustamujic A. Asthma and chronic obstructive pulmonary disease (COPD)—differences and similarities. Mater Sociomed. 2012;24:100–5.
Brightling CE, McKenna S, Hargadon B, Birring S, Green R, Siva R, et al. Sputum eosinophilia and the short term response to inhaled mometasone in chronic obstructive pulmonary disease. Thorax. 2005;60:193–8.
David B, Bafadhel M, Koenderman L, De Soyza A. Eosinophilic inflammation in COPD: from an inflammatory marker to a treatable trait. Thorax. 2021;76:188–95.
Benson VS, Hartl S, Barnes N, Galwey N, Van Dyke MK, Kwon N. Blood eosinophil counts in the general population and airways disease: a comprehensive review and meta-analysis. Eur Respir J. 2022. https://doi.org/10.1183/13993003.04590-2020.
Zhang S, King D, Rosen VM, Ismaila AS. Impact of single combination inhaler versus multiple inhalers to deliver the same medications for patients with asthma or COPD: a systematic literature review. Int J Chron Obstruct Pulmon Dis. 2020;15:417–38.
Pascoe S, Locantore N, Dransfield MT, Barnes NC, Pavord ID. Blood eosinophil counts, exacerbations, and response to the addition of inhaled fluticasone furoate to vilanterol in patients with chronic obstructive pulmonary disease: a secondary analysis of data from two parallel randomised controlled trials. Lancet Respir Med. 2015;3:435–42.
Wu C. Research progress of serum eosinophil in chronic obstructive pulmonary disease and asthma. Open Life Sci. 2023;18:20220779.
Smit JJ, Lukacs NW. A closer look at chemokines and their role in asthmatic responses. Eur J Pharmacol. 2006;533:277–88.
Saha S, Brightling CE. Eosinophilic airway inflammation in COPD. Int J Chron Obstruct Pulmon Dis. 2006;1:39–47.
Davoine F, Lacy P. Eosinophil cytokines, chemokines, and growth factors: emerging roles in immunity. Front Immunol. 2014;5:570.
Barnes PJ. Inflammatory endotypes in COPD. Allergy. 2019;74:1249–56.
Cook A, Harrington J, Simpson JL, Wark P. Mepolizumab asthma treatment failure due to refractory airway eosinophilia, which responded to benralizumab. Respirol Case Rep. 2021;9: e00743.
Gestal MC, Blas-Machado U, Johnson HM, Rubin LN, Dewan KK, Bryant C, et al. Disrupting immunosuppression reveals a role for eosinophils in coordinating the adaptive immune response in the respiratory tract. Microorganisms. 2020. https://doi.org/10.3390/microorganisms8111808.
Weissler JC. Eosinophilic lung disease. Am J Med Sci. 2017;354:339–49.
Siddiqui SH, Guasconi A, Vestbo J, Jones P, Agusti A, Paggiaro P, et al. Blood eosinophils: a biomarker of response to extrafine beclomethasone/formoterol in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2015;192:523–5.
Pavord ID, Korn S, Howarth P, Bleecker ER, Buhl R, Keene ON, et al. Mepolizumab for severe eosinophilic asthma (DREAM): a multicentre, double-blind, placebo-controlled trial. Lancet. 2012;380:651–9.
Spector SL, Tan RA. Is a single blood eosinophil count a reliable marker for “eosinophilic asthma?” J Asthma. 2012;49:807–10.
Roufosse F, Weller PF. Practical approach to the patient with hypereosinophilia. J Allergy Clin Immunol. 2010;126:39–44.
Lea S, Higham A, Beech A, Singh D. How inhaled corticosteroids target inflammation in COPD. Eur Respir Rev. 2023. https://doi.org/10.1183/16000617.0084-2023.
Singh D, Agusti A, Martinez FJ, Papi A, Pavord ID, Wedzicha JA, et al. Blood eosinophils and chronic obstructive pulmonary disease: a global initiative for chronic obstructive lung disease science committee 2022 review. Am J Respir Crit Care Med. 2022;206:17–24.
Ramakrishnan S, Jeffers H, Langford-Wiley B, Davies J, Thulborn SJ, Mahdi M, et al. Blood eosinophil-guided oral prednisolone for COPD exacerbations in primary care in the UK (STARR2): a non-inferiority, multicentre, double-blind, placebo-controlled, randomised controlled trial. Lancet Respir Med. 2024;12:67–77.
Southworth T, Beech G, Foden P, Kolsum U, Singh D. The reproducibility of COPD blood eosinophil counts. Eur Respir J. 2018. https://doi.org/10.1183/13993003.00427-2018.
Singh D, Kolsum U, Brightling CE, Locantore N, Agusti A, Tal-Singer R, et al. Eosinophilic inflammation in COPD: prevalence and clinical characteristics. Eur Respir J. 2014;44:1697–700.
Vedel-Krogh S, Nielsen SF, Lange P, Vestbo J, Nordestgaard BG. Blood eosinophils and exacerbations in chronic obstructive pulmonary disease. The Copenhagen General Population Study. Am J Respir Crit Care Med. 2016;193:965–74.
Bafadhel M, McKenna S, Terry S, Mistry V, Reid C, Haldar P, et al. Acute exacerbations of chronic obstructive pulmonary disease [internet]. Am J Respir Crit Care Med. 2011. https://doi.org/10.1164/rccm.201104-0597oc.
Hogg JC, Chu F, Utokaparch S, Woods R, Elliott WM, Buzatu L, et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med. 2004;350:2645–53.
Hastie AT, Martinez FJ, Curtis JL, Doerschuk CM, Hansel NN, Christenson S, et al. Association of sputum and blood eosinophil concentrations with clinical measures of COPD severity: an analysis of the SPIROMICS cohort. Lancet Respir Med. 2017;5:956–67.
Siva R, Green RH, Brightling CE, Shelley M, Hargadon B, McKenna S, et al. Eosinophilic airway inflammation and exacerbations of COPD: a randomised controlled trial. Eur Respir J. 2007;29:906–13.
Turato G, Semenzato U, Bazzan E, Biondini D, Tinè M, Torrecilla N, et al. blood eosinophilia neither reflects tissue eosinophils nor worsens clinical outcomes in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;197:1216–9.
Lacoste JY, Bousquet J, Chanez P, Van Vyve T, Simony-Lafontaine J, Lequeu N, et al. Eosinophilic and neutrophilic inflammation in asthma, chronic bronchitis, and chronic obstructive pulmonary disease. J Allergy Clin Immunol. 1993;92:537–48.
Jogdand P, Siddhuraj P, Mori M, Sanden C, Jönsson J, Walls AF, et al. Eosinophils, basophils and type 2 immune microenvironments in COPD-affected lung tissue. Eur Respir J. 2020. https://doi.org/10.1183/13993003.00110-2019.
Bateman ED, Reddel HK, van Zyl-Smit RN, Agusti A. The asthma–COPD overlap syndrome: towards a revised taxonomy of chronic airways diseases? Lancet Respir Med. 2015;3:719–28.
Hosseini M, Almasi-Hashiani A, Sepidarkish M, Maroufizadeh S. Global prevalence of asthma–COPD overlap (ACO) in the general population: a systematic review and meta-analysis. Respir Res. 2019;20:229.
Peng J, Wang M, Wu Y, Shen Y, Chen L. Clinical indicators for asthma–COPD overlap: a systematic review and meta-analysis. Int J Chron Obstruct Pulmon Dis. 2022;17:2567–75.
Bélanger M, Couillard S, Courteau J, Larivée P, Poder TG, Carrier N, et al. Eosinophil counts in first COPD hospitalizations: a comparison of health service utilization. Int J Chron Obstruct Pulmon Dis. 2018;13:3045–54.
Kostikas K, Papathanasiou E, Papaioannou AI, Bartziokas K, Papanikolaou IC, Antonakis E, et al. Blood eosinophils as predictor of outcomes in patients hospitalized for COPD exacerbations: a prospective observational study. Biomarkers. 2021;26:354–62.
Bartziokas K, Papathanasiou E, Papaioannou AI, Papanikolaou I, Antonakis E, Makou I, et al. Eosinopenia as a prognostic biomarker for noninvasive ventilation use in COPD exacerbations. J Pers Med. 2023. https://doi.org/10.3390/jpm13040686.
Bafadhel M, Greening NJ, Harvey-Dunstan TC, Williams JEA, Morgan MD, Brightling CE, et al. Blood eosinophils and outcomes in severe hospitalized exacerbations of COPD. Chest. 2016;150:320–8.
Liu H, Xie Y, Huang Y, Luo K, Gu Y, Zhang H, et al. The association between blood eosinophils and clinical outcome of acute exacerbations of chronic obstructive pulmonary disease: a systematic review and meta-analysis. Respir Med. 2024;222: 107501.
Shin SH, Park HY, Kang D, Cho J, Kwon SO, Park JH, et al. Serial blood eosinophils and clinical outcome in patients with chronic obstructive pulmonary disease. Respir Res. 2018;19:134.
Casanova C, Celli BR, de Torres JP, Martínez-Gonzalez C, Cosio BG, Pinto-Plata V, et al. Prevalence of persistent blood eosinophilia: relation to outcomes in patients with COPD. Eur Respir J. 2017. https://doi.org/10.1183/13993003.01162-2017.
Yun JH, Lamb A, Chase R, Singh D, Parker MM, Saferali A, et al. Blood eosinophil count thresholds and exacerbations in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2018;141:2037-47.e10.
Halpin DMG, Healey H, Skinner D, Carter V, Pullen R, Price D. Exacerbation history and blood eosinophil count prior to diagnosis of COPD and risk of subsequent exacerbations. Eur Respir J. 2024. https://doi.org/10.1183/13993003.02240-2023.
Petsky HL, Cates CJ, Lasserson TJ, Li AM, Turner C, Kynaston JA, et al. A systematic review and meta-analysis: tailoring asthma treatment on eosinophilic markers (exhaled nitric oxide or sputum eosinophils). Thorax. 2012;67:199–208.
Tran TN, Khatry DB, Ke X, Ward CK, Gossage D. High blood eosinophil count is associated with more frequent asthma attacks in asthma patients. Ann Allergy Asthma Immunol. 2014;113:19–24.
Pavord ID, Lettis S, Locantore N, Pascoe S, Jones PW, Wedzicha JA, et al. Blood eosinophils and inhaled corticosteroid/long-acting β-2 agonist efficacy in COPD. Thorax. 2016;71:118–25.
Barnes NC, Sharma R, Lettis S, Calverley PMA. Blood eosinophils as a marker of response to inhaled corticosteroids in COPD. Eur Respir J. 2016;47:1374–82.
Lipson DA, Barnhart F, Brealey N, Brooks J, Criner GJ, Day NC, et al. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378:1671–80.
Watz H, Tetzlaff K, Wouters EFM, Kirsten A, Magnussen H, Rodriguez-Roisin R, et al. Blood eosinophil count and exacerbations in severe chronic obstructive pulmonary disease after withdrawal of inhaled corticosteroids: a post-hoc analysis of the WISDOM trial. Lancet Respir Med. 2016;4:390–8.
Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, et al. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. N Engl J Med. 2016;374:2222–34.
Harries TH, Rowland V, Corrigan CJ, Marshall IJ, McDonnell L, Prasad V, et al. Blood eosinophil count, a marker of inhaled corticosteroid effectiveness in preventing COPD exacerbations in post-hoc RCT and observational studies: systematic review and meta-analysis. Respir Res. 2020;21:3.
Liu T, Xiang Z-J, Hou X-M, Chai J-J, Yang Y-L, Zhang X-T. Blood eosinophil count-guided corticosteroid therapy and as a prognostic biomarker of exacerbations of chronic obstructive pulmonary disease: a systematic review and meta-analysis. Ther Adv Chronic Dis. 2021;12:20406223211028770.
2024 GOLD Report [Internet]. Global initiative for chronic obstructive lung disease—GOLD. 2023. https://goldcopd.org/2024-gold-report/. Accessed Jul 11, 2024
Muro S, Kawayama T, Sugiura H, Seki M, Duncan EA, Bowen K, et al. Benefits of budesonide/glycopyrronium/formoterol fumarate dihydrate on lung function and exacerbations of COPD: a post-hoc analysis of the KRONOS study by blood eosinophil level and exacerbation history. Respir Res. 2024;25:297.
Mathioudakis AG, Bate S, Sivapalan P, Stæhr Jensen J-U, Singh D, Vestbo J. Rethinking blood eosinophils for assessing ICS response in COPD: a post hoc analysis from FLAME. Chest. 2024. https://doi.org/10.1016/j.chest.2024.06.3790.
Pavord ID, Lettis S, Anzueto A, Barnes N. Blood eosinophil count and pneumonia risk in patients with chronic obstructive pulmonary disease: a patient-level meta-analysis. Lancet Respir Med. 2016;4:731–41.
Aggarwal B, Jones P, Casas A, Gomes M, Juthong S, Litewka D, et al. Association between increased risk of pneumonia with ICS in COPD: a continuous variable analysis of patient factors from the IMPACT study. Pulm Ther. 2024;10:183–92.
Bafadhel M, McKenna S, Terry S, Mistry V, Pancholi M, Venge P, et al. Blood eosinophils to direct corticosteroid treatment of exacerbations of chronic obstructive pulmonary disease: a randomized placebo-controlled trial. Am J Respir Crit Care Med. 2012;186:48–55.
Serafino-Agrusa L, Scichilone N, Spatafora M, Battaglia S. Blood eosinophils and treatment response in hospitalized exacerbations of chronic obstructive pulmonary disease: a case-control study. Pulm Pharmacol Ther. 2016;37:89–94.
Bafadhel M, Davies L, Calverley PMA, Aaron SD, Brightling CE, Pavord ID. Blood eosinophil guided prednisolone therapy for exacerbations of COPD: a further analysis. Eur Respir J. 2014;44:789–91.
Sivapalan P, Lapperre TS, Janner J, Laub RR, Moberg M, Bech CS, et al. Eosinophil-guided corticosteroid therapy in patients admitted to hospital with COPD exacerbation (CORTICO-COP): a multicentre, randomised, controlled, open-label, non-inferiority trial. Lancet Respir Med. 2019;7:699–709.
Flood-Page P, Swenson C, Faiferman I, Matthews J, Williams M, Brannick L, et al. A study to evaluate safety and efficacy of mepolizumab in patients with moderate persistent asthma. Am J Respir Crit Care Med. 2007;176:1062–71.
Kips JC, O’Connor BJ, Langley SJ, Woodcock A, Kerstjens HAM, Postma DS, et al. Effect of SCH55700, a humanized anti-human interleukin-5 antibody, in severe persistent asthma: a pilot study. Am J Respir Crit Care Med. 2003;167:1655–9.
Ortega HG, Yancey SW, Mayer B, Gunsoy NB, Keene ON, Bleecker ER, et al. Severe eosinophilic asthma treated with mepolizumab stratified by baseline eosinophil thresholds: a secondary analysis of the DREAM and MENSA studies. Lancet Respir Med. 2016;4:549–56.
Laviolette M, Gossage DL, Gauvreau G, Leigh R, Olivenstein R, Katial R, et al. Effects of benralizumab on airway eosinophils in asthmatic patients with sputum eosinophilia. J Allergy Clin Immunol. 2013;132:1086-96.e5.
Varricchi G, Bagnasco D, Borriello F, Heffler E, Canonica GW. Interleukin-5 pathway inhibition in the treatment of eosinophilic respiratory disorders: evidence and unmet needs. Curr Opin Allergy Clin Immunol. 2016;16:186–200.
Pavord ID, Chanez P, Criner GJ, Kerstjens HAM, Korn S, Lugogo N, et al. Mepolizumab for eosinophilic chronic obstructive pulmonary disease. N Engl J Med. 2017;377:1613–29.
Brightling CE, Bleecker ER, Panettieri RA Jr, Bafadhel M, She D, Ward CK, et al. Benralizumab for chronic obstructive pulmonary disease and sputum eosinophilia: a randomised, double-blind, placebo-controlled, phase 2a study. Lancet Respir Med. 2014;2:891–901.
Criner GJ, Celli BR, Singh D, Agusti A, Papi A, Jison M, et al. Predicting response to benralizumab in chronic obstructive pulmonary disease: analyses of GALATHEA and TERRANOVA studies. Lancet Respir Med. 2020;8:158–70.
Trial [Internet]. https://cdek.pharmacy.purdue.edu/trial/NCT04098718/. Accessed Jul 12, 2024
Donovan T, Milan SJ, Wang R, Banchoff E, Bradley P, Crossingham I. Anti-IL-5 therapies for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2020;12:CD13432.
Rael EL, Lockey RF. Interleukin-13 signaling and its role in asthma. World Allergy Organ J. 2011;4:54–64.
Wenzel S, Castro M, Corren J, Maspero J, Wang L, Zhang B, et al. Dupilumab efficacy and safety in adults with uncontrolled persistent asthma despite use of medium-to-high-dose inhaled corticosteroids plus a long-acting β2 agonist: a randomised double-blind placebo-controlled pivotal phase 2b dose-ranging trial. Lancet. 2016;388:31–44.
Bhatt SP, Rabe KF, Hanania NA, Vogelmeier CF, Cole J, Bafadhel M, et al. Dupilumab for COPD with type 2 inflammation indicated by eosinophil counts. N Engl J Med. 2023;389:205–14.
Bhatt SP, Rabe KF, Hanania NA, Vogelmeier CF, Bafadhel M, Christenson SA, et al. Dupilumab for COPD with blood eosinophil evidence of type 2 inflammation. N Engl J Med. 2024;390:2274–83.
Liew FY, Girard J-P, Turnquist HR. Interleukin-33 in health and disease. Nat Rev Immunol. 2016;16:676–89.
Rabe KF, Celli BR, Wechsler ME, Abdulai RM, Luo X, Boomsma MM, et al. Safety and efficacy of itepekimab in patients with moderate-to-severe COPD: a genetic association study and randomised, double-blind, phase 2a trial. Lancet Respir Med. 2021;9:1288–98.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Konstantinos Bartziokas and Stelios Loukides: conceptualization, investigation, Konstantinos Bartziokas and Andriana I Papaioannou: writing—original draft, validation, Konstantinos Bartziokas, Nikoletta Rovina, Georgia Papaiakovou and Paschalis Steiropoulos writing—review & editing, supervision. All authors read and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of Interest
Paschalis Steiropoulos is an Editorial Board member of Pulmonary Therapy. Paschalis Steiropoulos was not involved in the selection of peer reviewers for the manuscript nor any of the subsequent editorial decisions. Konstantinos Bartziokas, Andriana I Papaioannou, Nikoletta Rovina, Georgia Loukides, and Stelios Loukides have nothing to disclose.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Bartziokas, K., Papaioannou, A.I., Rovina, N. et al. Εosinophilic Chronic Obstructive Pulmonary Disease. What Do We Know So Far?. Pulm Ther 11, 7–24 (2025). https://doi.org/10.1007/s41030-024-00280-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41030-024-00280-0