Abstract
Cadmium contamination in aquatic environments poses severe risks to aquatic organisms, particularly fish, where cadmium accumulation in tissues can lead to compromised organ functionality and reproductive issues. The present study aimed to assess the effects of cadmium (Cd) exposure on key biomarkers of oxidative stress, DNA damage, apoptosis, and enzyme activity in the liver and kidney tissues of rainbow trout (Oncorhynchus mykiss). Specifically, the study measured 8-hydroxy-2-deoxyguanosine (8-OHdG) levels, caspase-3 activation, acetylcholinesterase (AChE) activity, and oxidative stress indicators (ONOO−, MDA, GSH, SOD, and CAT) following exposure to three Cd concentrations (1, 3, and 5 mg/L) over three time points (24, 48, and 96 h). Tissue samples were collected post-exposure, and the analysis revealed a significant decrease in MDA levels in both tissues. GSH concentrations declined with prolonged exposure, while SOD activity increased, indicating a response to oxidative stress, contrasted by a reduction in CAT activity. An initial increase in ONOO− levels was observed at 24 h, followed by a subsequent decrease at the 48 and 96 h marks. These results suggest that cadmium induces oxidative stress in the liver and kidney tissues of fish. Cadmium exposure also significantly elevated 8-OHdG levels, signaling DNA damage, and increased caspase-3 activity, indicative of apoptosis, across all doses and time points (p < 0.05). The histological examination of liver and kidney showed tissue injury. Additionally, a negative correlation between AChE activity and exposure duration was noted, with prolonged exposure resulting in substantial AChE inhibition. Given the role of AChE in behavior regulation, these findings underscore the importance of exploring time-dependent, tissue-specific changes in AChE activity to further elucidate the mechanisms underlying cadmium-induced behavioral abnormalities.
Similar content being viewed by others
Introduction
The presence of various harmful compounds in the environment due to chemical pollution poses a risk to living organisms, exposing them to numerous hazardous substances over time. Many organisms inhabit regions where environmental pollution surpasses safe thresholds, with a significant portion attributed to the buildup of heavy metals. These metals are prevalent in numerous locations globally, particularly in proximity to industrial and urban centers1. Cadmium (Cd) is a highly toxic metal prevalent in the environment, entering aquatic ecosystems through both natural processes and anthropogenic activities, and posing significant risks to environmental and human health2. Aquatic organisms absorb cadmium primarily in its ionic form (Cd2⁺) directly from the water3. Exposure to cadmium can lead to several detrimental effects, including the inhibition of metal-dependent enzymes and the induction of oxidative stress. This stress arises from the generation of reactive oxygen species (ROS), such as superoxide anions (O2−) and hydroxyl radicals4,5. To counteract oxidative stress, aerobic organisms have developed sophisticated antioxidant defense mechanisms. These systems typically include a variety of antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT), glutathione reductase (GR), and glutathione peroxidase (GPx)6,7,8. Glutathione (GSH) also plays a critical role in this defense, acting as an antioxidant, enzyme cofactor, and the primary redox buffer. Although GSH is synthesized in most vertebrate tissues, the liver and kidney are particularly active sites for its production9,10. Cd can infiltrate the body via air, water, soil, and food. With a prolonged half-life ranging from 10 to 30 years, it predominantly accumulates in organs such as the liver, bones, and other tissues, leading to irreversible harm. The kidneys and liver are particularly vulnerable to the toxic effects of Cd. Cd exerts diverse effects on cellular functions, which vary depending on the specific cell type. It interferes with antioxidant defense systems, promotes the generation of reactive oxygen species (ROS), and increases the synthesis of nitric oxide (NO)11,12. One of the key oxidants produced as a result is peroxynitrite (ONOO−), a potent oxidizing agent that contributes to toxicity through mechanisms such as lipid peroxidation (LPO) and the depletion of GSH. Research in animal models has demonstrated that the liver and kidney are particularly vulnerable to chronic cadmium toxicity13.
Oxidative stress can induce DNA damage, generating various byproducts. Among these, 8-hydroxy-2ʹ-deoxyguanosine (8-OHdG), formed from the oxidation of guanine, is a crucial biomarker for detecting DNA injury. This modification occurs when hydroxyl radicals attack DNA, leading to the formation of 8-hydroxyguanine, which is subsequently converted to 8-OHdG14,15,16.
Caspases, a family of proteases, are central to programmed cell death or apoptosis. Initially produced as inactive precursors, they become activated through proteolytic cleavage. By dismantling proteins involved in DNA repair, caspases facilitate the release of endonucleases, enzymes that degrade DNA. Apoptosis serves as a cellular defense mechanism to eliminate DNA-damaged cells17,18,19.
Acetylcholinesterase (AChE) is another critical enzyme involved in various cellular processes, including apoptosis, cell communication, and proliferation. AChE is a crucial enzyme that plays a vital role in the nervous system of organisms. Due to its sensitivity to various pollutants, AChE has become a valuable tool in toxicological assessments. By measuring changes in AChE activity, scientists can detect the presence of harmful substances in the environment, particularly in aquatic ecosystems. A decline in AChE activity often indicates exposure to toxic chemicals, such as pesticides like glyphosate. This makes AChE a reliable biomarker for monitoring water quality and evaluating the overall health of aquatic environments. Researchers widely utilize AChE measurements, coupled with spectrophotometric analysis, to assess the impact of pollutants and inform water management strategies20,21,22,23,24.
The use of multiple biomarkers such as 8-OHdG, Caspase 3 Activation, AChE, and Antioxidant Enzyme Activity together in the study will allow us to obtain more comprehensive information about the different mechanisms triggered by cadmium in the fish body. In this way, not only the effects of cadmium on growth and development, but also more detailed mechanisms such as oxidative stress, apoptosis, and neurological damage can be examined. Histopathological examinations will support biochemical findings by visualizing the structural damage caused by cadmium in fish tissues and will provide a clearer picture. In this way, it can be better understood which organs and tissues are affected more by cadmium and what the long-term consequences of these effects may be. The originality of this study is revealed by the fact that there is no study in the current literature that examines the effects of cadmium on rainbow trout in such a comprehensive way and evaluates such a large number of biomarkers together.
Materials and methods
Chemicals and fish
Cadmium was purchased in a pure standard solution in concentration 1000 mg/L from Sigma-Aldrich (St. Louis, MO, USA). Rainbow trout (n = 216), with an average weight of 230.25 ± 12.10 gr., were obtained from a local aquaculture facility in Turkey. Upon receipt, the fish were relocated to the Fisheries Laboratory of the Faculty of Agriculture and were acclimated to laboratory settings in 600-L holding tanks for a period of two weeks. During this acclimatization phase, the fish were provided ad libitum with pelleted trout feed (Ecobio Inc.) twice daily. For the experimental procedures, three fish were used for each exposure duration, and each experiment was conducted in triplicate. Water circulation in the tanks was maintained at a flow rate of 1.5 L per minute. The physicochemical properties of the water, such as pH, dissolved oxygen, hardness, temperature, and alkalinity, were within the optimal range for fish survival25. Additionally, these parameters remained relatively stable, as shown in Supplementary Table 1.
Cadmium exposure
After a two-week acclimatization period, the fish were divided into two groups: one group served as a control and received no treatment, while the second group was exposed to a sub-lethal concentration fish were exposed to Cd doses of 1, 3, and 5 mg/L for 24, 48, and 96 h26. The lethal cadmium concentration for trout is 8 mg/L27. A stock solution of the anesthetic agent Tricaine methanesulfonate (MS-222) was prepared at a concentration of 10 g/L in 0.5 M NaHCO3 buffer (pH = 7.4). Fish were euthanized by placing them in 0.5 g/L anesthetic solution. Tissues were immediately frozen at − 80 °C after exposure for subsequent analysis. This study was executed in strict accordance with the ethical guidelines and protocols approved by the Bingöl University Animal Experiments Local Ethics Committee (2018/02–02/10), all the methods and results stated in the study were in accordance with ARRIVE guidelines and regulations.
Tissue preparation
Kidney and liver tissues from each fish were homogenized in physiological saline using a double-walled homogenizer kept in an ice bath. The homogenate was then centrifuged at 17,000g for 15 min at 4 °C (Micro Star 17R centrifuge, VWR) to analyze the supernatant. Protein concentrations were determined using the Folin reagent with bovine serum albumin as a standard28.
Biochemical assays
GSH measurement: Concentrations of total and reduced glutathione (GSH) were measured according to the method described by29.
Enzyme activities: CAT activity was evaluated using the method by30 and SOD activity was assessed following the protocol31. MDA concentrations, indicative of lipid peroxidation, were determined according to the method of32.
ONOO- production: ONOO- levels were quantitatively determined by diluting samples in 1.0 M NaOH and measuring absorbance increase at 302 nm.
AChE activity: AChE activity was determined using Ellman’s method33.
DNA damage: Levels of DNA damage were evaluated using the 8-OHdG ELISA kit (Beyotime Biotechnology).
Caspase-3 activity: Caspase-3 enzyme activity was measured using a commercial fish ELISA kit (Beyotime Biotechnology).
Histopathological examination
The livers, and kidneys were preserved in a chemical solution called formalin. Five micron thick sections of the sample tissues were obtained using rotary microtome (Leica RM 2125) and affixed to alcohol washed clean glass slides pre-coated with Mayor’s albumin and allowed to dry at room temperature for 12–16 h. The section was deparaffinised followed staining with Harris haematoxylin–eosin (HE) for histological studies and examined under a microscope (EclipseE100; Nikon, Tokyo, Japan). The damage to the tissues was assessed34.
Statistical analysis
Statistical evaluations were conducted using SPSS version 20.0. Results were expressed as mean ± standard error of the mean. A one-way ANOVA was employed to examine the impact of concentration and exposure time across various experimental groups. Duncan Test was utilized to ascertain the significance of differences between control and treatment groups at different concentrations and time intervals. Statistical significance was defined as p ≤ 0.05.
Results
In liver tissue exposed to cadmium for 24 h, MDA levels exhibited a statistically significant decrease compared to the control group, whereas a significant increase was observed in kidney tissue. However, after 48 and 96 h of exposure, while no statistically significant difference was observed between the control and 1 mg/L and 3 mg/L at 48 and 96 h of exposure, a significant difference occurred between the control and 5 mg/L in MDA levels were detected in liver tissue, a significant decrease was observed in kidney tissue (Fig. 1).
GSH concentrations showed no statistically significant change in the liver after 24 h of treatment compared to the control group, while in the kidney they increased significantly compared to the control group in all three groups after exposure. While there was no statistically significant difference between the control and 3 mg/L in the liver after 48 h of exposure, 1 mg/L and 5 mg/L showed a statistically significant decrease both among themselves and compared to the control group. In the kidney, only 3 mg/L showed a significant increase after 48 h of exposure. While a significant decrease was observed in both liver and kidney compared to the control group after 96 h of exposure, there was no significant difference between the doses (Fig. 2).
SOD activity was significantly elevated in both liver and kidney tissues after 24 h of treatment compared to the control group. However, no statistically significant changes were observed in SOD activity in either tissue after 48 h relative to the control group. At the 96-h mark, a significant increase in SOD activity was noted in liver tissue only at the 3 mg/L cadmium dose. Conversely, a significant decrease in SOD activity was observed in kidney tissue at the 1 mg/L dose level (Fig. 3).
CAT activity significantly increased in both liver and kidney tissues following 24 h of treatment compared to the control group. After 48 h, no significant changes in CAT activity were detected in kidney tissue relative to the control group, whereas a significant decrease was observed in liver tissue at the 3 mg/L dose level. Following 96 h of treatment, liver and kidney tissues exhibited a significant reduction in CAT activity at the 3 and 5 mg/L dose levels compared to the control group (Fig. 4).
ONOO− levels significantly increased in both liver and kidney tissues after 24 h of treatment compared to the control group. After 48 h, a significant increase in ONOO− levels was observed in liver tissue only at the 5 mg/L dose level, while no significant changes were detected in kidney tissue. Following 96 h of treatment, liver tissue exhibited a significant decrease in ONOO− levels at the 1 mg/L dose level compared to the control group, whereas kidney tissue showed a significant decrease at the 3 and 5 mg/L dose levels relative to the control group (Fig. 5).
While there was no significant change in AChE activity in the liver between the control and 1 mg/L dose at 24-h exposure, a significant decrease was observed at doses 3 mg/L and 5 mg/L. At 48 and 96 h of exposure, there was a statistically significant difference between both control groups and doses. In the kidney, at 24-h exposure, there was a significant decrease between the control, 1 mg/L and 3 mg/L doses, while the 5 mg/L dose showed an insignificant decrease with the control, it showed a greater increase than the 1 mg/L and 3 mg/L doses. At 48 and 96-h exposure, a significant difference was observed only between the 3 mg/L dose and the other dose and the control group. (Fig. 6).
The levels of 8-OHdG exhibited a statistically significant increase in fish tissues relative to the control group, with variations dependent on both the duration of exposure and the dose administered (Fig. 7).
Caspase-3 activity In liver and kidney at 24-h exposure, no significant difference was observed between control and 1 mg/L dose, while a statistically significant increase was observed between control and 3 mg/L and 5 mg/L doses. In liver at 48-h exposure, no significant difference was observed between control and 1 dose, while a statistically significant decrease was observed between control and 3 mg/L. and 5 mg/L doses. In kidney at 48 h, doses caused a significant increase compared to control. In liver and kidney at 96-h exposure, no significant difference was observed between control and 1 mg/L dose, while a statistically significant increase was observed between control and 3 mg/L and 5 mg/L doses. (Fig. 8).
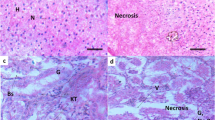
The histopathological examination of the liver and kidney of control and exposed Rainbow trout is presented in Fig. 9. In the liver tissue, the control group exhibited normal hepatocytes and nuclei (Fig. 9a), while the exposed group displayed congestion of blood vessels and necrosis (Fig. 9 b). These findings suggest that an excessive amount of heavy metals leads to histological alterations primarily through the production of reactive oxygen species. Histopathological examinations of liver and kidney of reference and exposed rainbow trout are shown in Fig. 9. In liver tissue, reference fish showed normal hepatocytes and nuclei (Fig. 9a), whereas exposed fish showed congestion of blood vessels and necrosis (Fig. 9b). Kidney sections of the reference group shown in Fig. 9 c showed a normal structure, including smooth renal tubules and Bowman’s space. Conversely, the primary distinguishing characteristics observed in the exposed fish tissue were a reduced or absent Bowman’s space, necrosis, and damaged tubules (Fig. 9d).
Discussion
Cadmium exposure has been extensively studied across different fish species, revealing varied toxic effects on antioxidant defenses depending on factors such as developmental stage, tissue type, exposure medium, and both concentration and duration of exposure. Cd, a metal lacking redox potential, primarily induces oxidative damage by disrupting antioxidant defenses, particularly those involving thiol-containing antioxidants and enzymes35. Environmental pollutants are known to induce oxidative stress through the production of ROS36. An imbalance between antioxidant and prooxidant systems can result in oxidative stress, causing ROS to damage nucleic acids, lipids, and proteins37. Oxidative stress results from an imbalance between oxidants and antioxidants, with an increase in reactive oxygen species (ROS) leading to oxidative damage38,39. Highly reactive free radicals can interact with unsaturated fatty acids in cell membranes, initiating lipid peroxidation and resulting in secondary products such as MDA40,41. Notably, MDA levels decreased significantly in exposed fish compared to controls, which is often linked to heightened antioxidant activity that reduces oxidative by-products. Although this hypothesis is plausible, it remains unproven, despite some studies reporting significant correlations between enhanced antioxidant defenses and reduced MDA levels in aquatic species42,43.
Oxidative damage is associated with ROS formation44 and occurs when antioxidant and detoxification systems fail to neutralize active intermediates from xenobiotics and their metabolites. Lipid peroxidation serves as a critical marker of oxidative damage to cellular components. Key antioxidant enzymes such as SOD, CAT, and GSH play crucial roles in mitigating oxidative stress45. Increased activity of these enzymes indicates oxidative stress. SOD, a group of metalloenzymes, is a primary antioxidant, while CAT, found in peroxisomes, converts hydrogen peroxide into water and molecular oxygen. In this study, SOD activity increased, whereas CAT activity generally decreased. The rise in SOD activity suggests that superoxide anion radicals are being produced, leading to a compensatory increase in antioxidant defenses46. This pattern aligns with typical responses to oxidative stress, where SOD activity rises to manage ROS levels, as observed in Japanese flounder (Paralichthys olivaceus) larvae, Mozambique tilapia, and gibel carp47,48,49. GSH is an essential endogenous antioxidant that protects cells from oxidative damage by interacting with free radicals and peroxides and maintaining protein sulfhydryl groups in their reduced state50,51.
ONOO−, formed from the reaction between NO and O2−, is a significant biological oxidant. While ONOOˉ formation is usually minimal, it can rise dramatically under conditions where ONOOˉ and O2ˉ levels are elevated or SOD activity is reduced, which often occurs in pathological states. ONOOˉ can not only initiate radical reactions but also cause nitration of biomolecules52. The study data revealed a significant increase in ONOO− levels at 24 h, followed by a decrease at 48 and 94 h, indicating an initial response of tissues to cadmium exposure.
Caspases are critical enzymes involved in apoptosis, a regulated process essential for tissue development, homeostasis, and immune responses. Excessive oxidative stress, often induced by environmental pollutants, can trigger apoptosis by disrupting antioxidant defenses53,54. The significant increase in caspase-3 activity observed in fish tissues suggests heightened apoptosis, likely a result of oxidative stress leading to cellular damage and dysfunction.
AChE, found predominantly at nerve-muscle junctions and synapses, is essential for terminating nerve impulses by breaking down acetylcholine into choline and acetic acid55. AChE is crucial for various physiological functions, including predator avoidance and prey location56. The observed decrease in AChE activity in all examined tissues aligns with findings from previous studies, which reported changes in AChE activity in response to environmental pollutants24,57,58,59. These alterations in AChE activity may have significant implications for fish health and behavior.
DNA is a primary target for oxidative damage, with 8-OHdG serving as a key biomarker for assessing such damage60,61. Exposure to pollutants, including pesticides, can induce oxidative stress and elevate 8-OHdG levels, indicating DNA damage62,63. This study observed increased 8-OHdG activity in all tissues examined, suggesting a correlation between oxidative damage and DNA damage64,65. The direct effect of MDA on DNA, combined with the correlation between 8-OHdG and MDA, suggests a link between lipid peroxidation and 8-OHdG formation. The data from this study on MDA aligns with the findings for 8-OHdG, supporting the hypothesis that the relationship between 8-OHdG and MDA can explain the contribution of lipid peroxidation to DNA damage66,67.
The study revealed that control fish exhibited normal hepatocytes and nuclei, while exposed fish displayed obstructed blood vessels and necrosis. These findings suggest that an excessive amount of heavy metals leads to histological alterations primarily through the production of reactive oxygen species. Previous research has documented hepatocellular damage caused by heavy metal overload in other fish species68,69,70,71,72. Similarly, in the kidney sections, while the control group demonstrated normal structures like regular renal tubules and Bowman’s space, the findings of decreased or absent Bowman’s space, necrosis, and damaged tubules in the exposed fish align with the results of other studies examining the kidney structures of fish exposed to metal-contaminated water. These studies have reported similar effects, such as a reduced renal hematopoietic system, tissue damage, necrosis, glomerular injury, proliferation of connective tissue, and narrowing of glomerular and epithelial tubules70,73,74,75. These results demonstrate consistent organ-specific damage patterns that expand our understanding of heavy metal toxicity in aquatic species.
Conclusion
The study’s significance lies in its comprehensive assessment of cadmium toxicity in rainbow trout, providing valuable insights into the mechanisms of heavy metal-induced organ damage and offering crucial information for environmental monitoring and management. These findings provide valuable insights into the molecular mechanisms underlying cadmium toxicity and form a foundation for future ecotoxicological research. To better understand the complex interactions between oxidative stress and observed biochemical and physiological responses, further studies are recommended on various aquatic organisms under prolonged cadmium exposure.
Data availability
The data produced in this study can be obtained from the corresponding author upon reasonable request.
References
Zwolak, A., Sarzyńska, M., Szpyrka, E. & Stawarczyk, K. Sources of soil pollution by heavy metals and their accumulation in vegetables: A review. Water Air Soil Pollut. 230, 1–9. https://doi.org/10.1007/s11270-019-4221-y (2019).
Faroon, O. et al. Toxicological profile for cadmium. (2013).
Goyer, R. A., Miller, C. R., Zhu, S.-Y. & Victery, W. Non-metallothionein-bound cadmium in the pathogenesis of cadmium nephrotoxicity in the rat. Toxicol. Appl. Pharmacol. 101, 232–244. https://doi.org/10.1016/0041-008X(89)90272-X (1989).
Nordberg, G. F., Nogawa, K., Nordberg, M. & Friberg, L. T. Handbook on the Toxicology of Metals (Third Edition) (eds Nordberg, G. F. et al.) 445–486. https://doi.org/10.1016/B978-012369413-3/50078-1 (Academic Press, 2007).
Kirici, M., Atamanalp, M., Kirici, M. & Beydemir, S. In vitro effects of some metal ions on glutathione reductase in the gills and liver of Capoeta trutta. Regul. Mech. Biosyst. 8, 66–70. https://doi.org/10.15421/021712 (2017).
Bakr, Z. et al. Toxicity of silver, copper oxide, and polyethylene nanoparticles on the earthworm Allolobophora caliginosa using multiple biomarkers. Appl. Soil. Ecol. 181, 104681. https://doi.org/10.1016/j.apsoil.2022.104681 (2023).
Soliman, H. A., Hamed, M. & Sayed, A.E.-D.H. Investigating the effects of copper sulfate and copper oxide nanoparticles in Nile tilapia (Oreochromis niloticus) using multiple biomarkers: the prophylactic role of Spirulina. Environ. Sci. Pollut. Res. 28, 30046–30057. https://doi.org/10.1007/s11356-021-12859-0 (2021).
Kirici, M., Kirici, M., Beydemir, S. & Atamanalp, M. Purification of carbonic anhydrase from Capoeta umbla (Heckel, 1843) gills and toxicological effects of some metals on enzyme activity. Turk. J. Fish Aquat. Sc. 16, 169–175. https://doi.org/10.4194/1303-2712-v16_1_17 (2016).
Dringen, R., Gutterer, J. M. & Hirrlinger, J. Glutathione metabolism in brain: metabolic interaction between astrocytes and neurons in the defense against reactive oxygen species. Eur. J. Biochem. 267, 4912–4916. https://doi.org/10.1046/j.1432-1327.2000.01597.x (2000).
Kırıcı, M., Kırıcı, M., Atamanalp, M. & Beydemir, Ş. Purification of glutathione reductase from some tissues of Capoeta umbla and the inhibitory effects of some metal ions on enzyme activity. Mar. Sci. Technol. Bull. 10, 193–200. https://doi.org/10.33714/masteb.769454 (2021).
Joseph, P. Mechanisms of cadmium carcinogenesis. Toxicol. Appl. Pharmacol. 238, 272–279. https://doi.org/10.1016/j.taap.2009.01.011 (2009).
Yang, H. & Shu, Y. Cadmium transporters in the kidney and cadmium-induced nephrotoxicity. Int. J. Mol. Sci. 16, 1484–1494. https://doi.org/10.3390/ijms16011484 (2015).
Bove, P. F. & van der Vliet, A. Nitric oxide and reactive nitrogen species in airway epithelial signaling and inflammation. Free Radic. Biol. Med. 41, 515–527. https://doi.org/10.1016/j.freeradbiomed.2006.05.011 (2006).
Topal, A., Oğuş, H., Sulukan, E., Comaklı, S. & Ceyhun, S. B. Okadaic acid enhances NfKB, TLR-4, caspase 3, ERK ½, c-FOS, and 8-OHdG signaling pathways activation in brain tissues of zebrafish larvae. Fish Shellfish Immun. 149, 109529. https://doi.org/10.1016/j.fsi.2024.109529 (2024).
Liao, G. et al. Urinary metals and their associations with DNA oxidative damage among e-waste recycling workers in Hong Kong. Ecotoxicol. Environ. Saf. 284, 116872. https://doi.org/10.1016/j.ecoenv.2024.116872 (2024).
Temiz, Ö. & Kargın, D. Physiological responses of oxidative damage, genotoxicity and hematological parameters of the toxic effect of neonicotinoid-thiamethoxam in Oreochromis niloticus. Environ. Toxicol. Phar. 106, 104377. https://doi.org/10.1016/j.etap.2024.104377 (2024).
Reyes-Becerril, M. et al. Caspase -1, -3, -8 and antioxidant enzyme genes are key molecular effectors following Vibrio parahaemolyticus and Aeromonas veronii infection in fish leukocytes. Immunobiology 223, 562–576. https://doi.org/10.1016/j.imbio.2018.07.002 (2018).
Yu, J. et al. Effects of nitrate (NO3−) stress-induced exacerbated cadmium (Cd2+) toxicity on the inflammatory response, oxidative defense, and apoptosis in juvenile Japanese flounder (Paralichthys olivaceus). J. Environ. Sci. 152, 535–548. https://doi.org/10.1016/j.jes.2024.05.036 (2025).
Liang, J. et al. Molecular mechanism of apoptosis induced by 4-tBP in common carp (Cyprinus carpio L.) head kidneys was explored from various angles: Hippo pathway, miR-203a, oxidative stress, ER stress, and mitochondrial pathway. Aquaculture 589, 740981. https://doi.org/10.1016/j.aquaculture.2024.740981 (2024).
Hernández Díaz, M. et al. Polluted water from a storage dam (Villa Victoria, méxico) induces oxidative damage, AChE activity, embryotoxicity, and behavioral changes in Cyprinus carpio larvae. Environ. Res. 258, 119282. https://doi.org/10.1016/j.envres.2024.119282 (2024).
Hamed, M., Martyniuk, C. J., Soliman, H. A. M., Osman, A. G. M. & Said, R. E. M. Neurotoxic and cardiotoxic effects of pyrogallol on catfish (Clarias gariepinus). Environ. Toxicol. Phar. 109, 104481. https://doi.org/10.1016/j.etap.2024.104481 (2024).
Sharma, G. & Chadha, P. Toxic effects of aniline in liver, gills and kidney of freshwater fish Channa punctatus after acute exposure. Compar. Biochem. Physiol. Part C Toxicol. Pharmacol. 281, 109916. https://doi.org/10.1016/j.cbpc.2024.109916 (2024).
Thanomsit, C. et al. The glyphosate (Roundup): fate in aquatic environment, adverse effect and toxicity assessment in aquatic organisms. Naresuan Univ. J. Sci. Technol. 28, 65–81 (2020).
Kirici, M. Assessment of 8-hydroxy-2-deoxyguanosine activity, apoptosis, acetylcholinesterase and antioxidant enzyme activity in brain exposed to chlorpyrifos. Oceanol. Hydrobiol. Stud. 51, 167–177. https://doi.org/10.26881/oandhs-2022.2.05 (2022).
Rice, E. W., Bridgewater, L. & Association, A. P. H. Standard Methods for the Examination of Water and Wastewater Vol. 10 (American Public Health Association, 2012).
Hisar, O. et al. Kinetic behaviour of glucose 6-phosphate dehydrogenase and 6-phosphogluconate dehydrogenase in different tissues of rainbow trout (Oncorhynchus mykiss) exposed to non-lethal concentrations of cadmium. Acta Vet. Brno 78, 179–185. https://doi.org/10.2754/avb200978010179 (2009).
Kayhan, F. E. Su Ürünlerinde Kadmiyumun Biyobirikimi ve Toksisitesi. Ege J. Fish. Aquat. Sci. 23, 215–220 (2006).
Lowry, O. H., Rosebrough, N. J., Farr, A. L. & Randall, R. J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275. https://doi.org/10.1016/S0021-9258(19)52451-6 (1951).
Sedlak, J. & Lindsay, R. H. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal. Biochem. 25, 192–205. https://doi.org/10.1016/0003-2697(68)90092-4 (1968).
Aebi, H. Methods of enzymatic analysis 673–684. https://doi.org/10.1016/B978-0-12-091302-2.50032-3 (Elsevier, 1974).
Sun, Y., Oberley, L. W. & Li, Y. A simple method for clinical assay of superoxide dismutase. Clin. Chem. 34, 497–500. https://doi.org/10.1093/clinchem/34.3.497 (1988).
Ohkawa, H., Ohishi, N. & Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 95, 351–358. https://doi.org/10.1016/0003-2697(79)90738-3 (1979).
Ellman, G. L., Courtney, K. D., Andres, V. Jr. & Featherstone, R. M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95. https://doi.org/10.1016/0006-2952(61)90145-9 (1961).
Topal, A., Oruç, E., Altun, S., Ceyhun, S. B. & Atamanalp, M. The effects of acute boric acid treatment on gill, kidney and muscle tissues in juvenile rainbow trout. J. Appl. Anim. Res. 44, 297–302. https://doi.org/10.1080/09712119.2015.1031784 (2016).
Wang, N. et al. Effect on intestinal microbiota, bioaccumulation, and oxidative stress of Carassius auratus gibelio under waterborne cadmium exposure. Fish Physiol. Biochem. 46, 2299–2309. https://doi.org/10.1007/s10695-020-00870-0 (2020).
Ferreira, M., Moradas-Ferreira, P. & Reis-Henriques, M. Oxidative stress biomarkers in two resident species, mullet (Mugil cephalus) and flounder (Platichthys flesus), from a polluted site in River Douro Estuary, Portugal. Aquat. Toxicol. 71, 39–48. https://doi.org/10.1016/j.aquatox.2004.10.009 (2005).
Topal, A., Atamanalp, M., Oruç, E. & Erol, H. S. Physiological and biochemical effects of nickel on rainbow trout (Oncorhynchus mykiss) tissues: assessment of nuclear factor kappa B activation, oxidative stress and histopathological changes. Chemosphere 166, 445–452. https://doi.org/10.1016/j.chemosphere.2016.09.106 (2017).
Su, L. et al. Parental exposure to environmental cadmium causes developmental defects in the rare minnow (Gobiocypris rarus) embryos: The association between ROS-mediated oxidative stress and developmental dysfunction. J. Environ. Chem. Eng. 11, 111264. https://doi.org/10.1016/j.jece.2023.111264 (2023).
Sevcikova, M., Modra, H., Slaninova, A. & Svobodova, Z. Metals as a cause of oxidative stress in fish: a review. https://doi.org/10.17221/4272-VETMED (2011).
Wu, C., Zhang, Y., Chai, L. & Wang, H. Oxidative stress, endocrine disruption, and malformation of Bufo gargarizans embryo exposed to sub-lethal cadmium concentrations. Environ. Toxicol. Phar. 49, 97–104. https://doi.org/10.1016/j.etap.2016.12.005 (2017).
Yin, Y. et al. Amelioration of Cd-induced oxidative stress, MT gene expression, and immune damage by vitamin C in grass carp kidney cells. Biol. Trace Elem. Res. 194, 552–559. https://doi.org/10.1007/s12011-019-01808-1 (2020).
Ferreira, C. P. et al. Metal bioaccumulation, oxidative stress and antioxidant responses in oysters Crassostrea gasar transplanted to an estuary in southern Brazil. Sci. Total Environ. 685, 332–344. https://doi.org/10.1016/j.scitotenv.2019.05.384 (2019).
Silva, D. C. et al. Effects of parabens on antioxidant system and oxidative damages in Nile tilapia (Oreochromis niloticus). Ecotoxicol. Environ. Saf. 162, 85–91. https://doi.org/10.1016/j.ecoenv.2018.06.076 (2018).
Karaca, M. et al. Organochlorine pesticides and antioxidant enzymes are inversely correlated with liver enzyme gene expression in Cyprinus carpio. Toxicol. Lett. 230, 198–207. https://doi.org/10.1016/j.toxlet.2014.02.013 (2014).
Lei, X. G. et al. Paradoxical roles of antioxidant enzymes: basic mechanisms and health implications. Physiol. Rev. 96, 307–364. https://doi.org/10.1152/physrev.00010.2014 (2016).
Oruc, E. Oxidative stress responses and recovery patterns in the liver of Oreochromis niloticus exposed to chlorpyrifos-ethyl. Bull. Environ. Contam. Toxicol. 88, 678–684. https://doi.org/10.1007/s00128-012-0548-4 (2012).
Lee, D.-C., Choi, Y. J. & Kim, J.-H. Toxic effects of waterborne cadmium exposure on hematological parameters, oxidative stress, neurotoxicity, and heat shock protein 70 in juvenile olive flounder, Paralichthys olivaceus. Fish Shellfish Immun. 122, 476–483. https://doi.org/10.1016/j.fsi.2022.02.022 (2022).
Basha, P. S. & Rani, A. U. Cadmium-induced antioxidant defense mechanism in freshwater teleost Oreochromis mossambicus (Tilapia). Ecotoxicol. Environ. Saf. 56, 218–221. https://doi.org/10.1016/S0147-6513(03)00028-9 (2003).
Shi, H., Sui, Y., Wang, X., Luo, Y. & Ji, L. Hydroxyl radical production and oxidative damage induced by cadmium and naphthalene in liver of Carassius auratus. Compar. Biochem. Physiol. Part C Toxicol. Pharmacol. 140, 115–121. https://doi.org/10.1016/j.cca.2005.01.009 (2005).
Srikanth, K., Pereira, E., Duarte, A. & Ahmad, I. Glutathione and its dependent enzymes’ modulatory responses to toxic metals and metalloids in fish—a review. Environ. Sci. Pollut. Res. 20, 2133–2149. https://doi.org/10.1007/s11356-012-1459-y (2013).
Birben, E., Sahiner, U. M., Sackesen, C., Erzurum, S. & Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 5, 9–19. https://doi.org/10.1097/WOX.0b013e3182439613 (2012).
Valko, M., Rhodes, C. J., Moncol, J., Izakovic, M. & Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chemico-Biol. Interact. 160, 1–40. https://doi.org/10.1016/j.cbi.2005.12.009 (2006).
Teng, M. et al. Chronic toxic effects of flutolanil on the liver of zebrafish (Danio rerio). Chem. Res. Toxicol. 32, 995–1001. https://doi.org/10.1021/acs.chemrestox.8b00300 (2019).
Dumétier, B., Zadoroznyj, A. & Dubrez, L. IAP-mediated protein ubiquitination in regulating cell signaling. Cells https://doi.org/10.3390/cells9051118 (2020).
Olivares-Rubio, H. F. & Espinosa-Aguirre, J. J. Acetylcholinesterase activity in fish species exposed to crude oil hydrocarbons: A review and new perspectives. Chemosphere 264, 128401. https://doi.org/10.1016/j.chemosphere.2020.128401 (2021).
Berman, H. A., Yguerabide, J. & Taylor, P. Fluorescence energy transfer on acetylcholinesterase: spatial relationship between peripheral site and active center. Biochemistry 19, 2226–2235. https://doi.org/10.1021/bi00551a036 (1980).
Cesarino, I., Moraes, F. C., Lanza, M. R. & Machado, S. A. Electrochemical detection of carbamate pesticides in fruit and vegetables with a biosensor based on acetylcholinesterase immobilised on a composite of polyaniline–carbon nanotubes. Food Chem. 135, 873–879. https://doi.org/10.1016/j.foodchem.2012.04.147 (2012).
Kim, J.-H., Kim, S. K. & Hur, Y. B. Toxic effects of waterborne nitrite exposure on antioxidant responses, acetylcholinesterase inhibition, and immune responses in olive flounders, Paralichthys olivaceus, reared in bio-floc and seawater. Fish Shellfish Immun. 97, 581–586. https://doi.org/10.1016/j.fsi.2019.12.059 (2020).
Tripathi, A. et al. Assessment of triclosan induced histopathological and biochemical alterations, and molecular docking simulation analysis of acetylcholinesterase enzyme in the gills of fish, Cyprinus carpio. Environm. Sci. Pollut. Res. https://doi.org/10.1007/s11356-024-33840-7 (2024).
Thompson, H. J. et al. Effect of increased vegetable and fruit consumption on markers of oxidative cellular damage. Carcinogenesis 20, 2261–2266. https://doi.org/10.1093/carcin/20.12.2261 (1999).
Zhang, X., Wu, R. S., Fu, W., Xu, L. & Lam, P. K. Production of reactive oxygen species and 8-hydroxy-2′ deoxyguanosine in KB cells co-exposed to benzo [a] pyrene and UV-A radiation. Chemosphere 55, 1303–1308. https://doi.org/10.1016/j.chemosphere.2003.12.004 (2004).
Alak, G. et al. Antioxidant potential of ulexite in zebrafish brain: assessment of oxidative DNA damage, apoptosis, and response of antioxidant defense system. Biol. Trace Elem. Res. 199, 1092–1099. https://doi.org/10.1007/s12011-020-02231-7 (2021).
Atamanalp, M. et al. Treatment of oxidative stress, apoptosis, and DNA injury with N-acetylcysteine at simulative pesticide toxicity in fish. Toxicol. Mech. Methods 31, 224–234. https://doi.org/10.1080/15376516.2021.1871794 (2021).
Topal, A., Alak, G., Altun, S., Erol, H. S. & Atamanalp, M. Evaluation of 8-hydroxy-2-deoxyguanosine and NFkB activation, oxidative stress response, acetylcholinesterase activity, and histopathological changes in rainbow trout brain exposed to linuron. Environ. Toxicol. Phar. 49, 14–20. https://doi.org/10.1016/j.etap.2016.11.009 (2017).
Anjana Vaman, V., Tinu, S., Geetha, C., Lissy, K. & Mohanan, P. Effect of fibrin glue on antioxidant defense mechanism, oxidative DNA damage and chromosomal aberrations. Toxicol. Mech. Methods 23, 500–508. https://doi.org/10.3109/15376516.2013.785625 (2013).
Marnett, L. J. Oxyradicals and DNA damage. Carcinogenesis 21, 361–370. https://doi.org/10.1093/carcin/21.3.361 (2000).
Wong, Y. T., Ruan, R. & Tay, F. E. H. Relationship between levels of oxidative DNA damage, lipid peroxidation and mitochondrial membrane potential in young and old F344 rats. Free Radic. Res. 40, 393–402. https://doi.org/10.1080/10715760600556074 (2006).
Zheng, J.-L. et al. Effect of waterborne zinc exposure on metal accumulation, enzymatic activities and histology of Synechogobius hasta. Ecotoxicol. Environ. Saf. 74, 1864–1873. https://doi.org/10.1016/j.ecoenv.2011.06.018 (2011).
Javed, M., Ahmad, M. I., Usmani, N. & Ahmad, M. Multiple biomarker responses (serum biochemistry, oxidative stress, genotoxicity and histopathology) in Channa punctatus exposed to heavy metal loaded waste water. Sci. Rep. 7, 1675. https://doi.org/10.1038/s41598-017-01749-6 (2017).
Al-Bairuty, G. A., Shaw, B. J., Handy, R. D. & Henry, T. B. Histopathological effects of waterborne copper nanoparticles and copper sulphate on the organs of rainbow trout (Oncorhynchus mykiss). Aquat. Toxicol. 126, 104–115. https://doi.org/10.1016/j.aquatox.2012.10.005 (2013).
Fatima, A. et al. Ameliorating effects of natural herbal supplements against water-borne induced toxicity of heavy metals on Nile tilapia, (Oreochromis niloticus). Sci. Rep. 14, 22571. https://doi.org/10.1038/s41598-024-72268-4 (2024).
Singh, S. et al. Oxidative stress, inflammation, and steatosis elucidate the complex dynamics of HgCl2 induced liver damage in Channa punctata. Sci. Rep. 14, 9161. https://doi.org/10.1038/s41598-024-59917-4 (2024).
Abdel-Khalek, A. Risk assessment, bioaccumulation of metals and kidney of Leuciscus cephalus following exposure to heavy metals in the Tur River, North Western Romania. Ecotoxicol. Environ. Saf. 119, 198–205. https://doi.org/10.1016/j.ecoenv.2015.05.029 (2015).
Triastuti, J., Luthfiyah, L. & Satriya, M. G. R. I. In IOP Conference Series: Earth and Environmental Science. 012017 (IOP Publishing).
Chijioke, F. O. N., Chinedu, N., Benjamin, A. & JC, I. Toxicity of mancozeb on the African catfish Clarias gariepinus juveniles kidney, liver and spleen histoarchitecture. J. Clin. Toxicol. 14, 555 (2024).
Author information
Authors and Affiliations
Contributions
Mehmet Reşit Taysı: planned, analyzed, reviewed, wrote and edited the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All experimental protocols of this study were approved by the Bingöl University Animal Experiments Local Ethics Committee with the approval number of meetings 2018/02 and number of decisions 02/10. The study was conducted in conformity with the ARRIVE.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Taysı, M.R. Assessing the effects of cadmium on antioxidant enzymes and histological structures in rainbow trout liver and kidney. Sci Rep 14, 27453 (2024). https://doi.org/10.1038/s41598-024-78835-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-78835-z