Abstract
Subarachnoid hemorrhage (SAH) occurs when blood enters the subarachnoid space, typically due to aneurysm rupture, triggering complex pathophysiological processes. A reliable animal model is crucial for simulating SAH and investigating mechanisms of brain white matter injury. SAH was induced in C57BL/6J mice using an intraluminal perforation technique. Various filaments were tested to determine the optimal one, and filament depth was carefully measured. Postoperative evaluations included monitoring body weight, blood distribution on the skull, and clot formation. Cerebral blood flow was assessed, and neurological function was evaluated using modified Garcia scores, open field tests, and gait analysis. Myelin integrity was assessed by Luxol fast blue staining, and immunofluorescence was used to examine myelin, microglia, and neuronal integrity in the cortex and striatum. Using 4 − 0 polypropylene filaments advanced to 13 ± 1 mm at a 15–20 degree, we established a stable SAH mouse model with a success rate of 91.43% and a mortality rate of 6.25%. The SAH group showed motor impairments at 48 h post-surgery, along with myelin damage in the corpus callosum and striatum, oligodendrocyte damage, and neuronal injury.Our improved intraluminal perforation technique offers a stable and standardized SAH model, providing a reliable platform for studying SAH pathophysiology and testing new therapies.
Similar content being viewed by others
Introduction
Subarachnoid hemorrhage (SAH) occurs when arterial blood enters the subarachnoid space, typically affecting the Circle of Willis and its branches, leading to significant neurological damage and impaired function with poor prognosis1,2. Researchers have developed various experimental models of SAH, including commonly used vascular perforation models in rats and mice3,4, single or repeated injections of autologous blood, red blood cells, or thrombin into the cisterna magna or optic chiasm5,6, and direct targeted intracranial vascular punctures.
While these models replicate certain pathological processes of SAH, they have inherent limitations. For instance, the blood or red cell injection models effectively simulate the pathogenic effects of red blood cells in cerebrospinal fluid but are limited by the small injection volume and single-component nature, which fail to reproduce the sustained pathological changes observed in SAH7. Additionally, intracranial targeted puncture represents an invasive, open-cranial procedure that disrupts skull integrity, affecting the enclosed intracranial environment8,9.
Due to the lower genetic homology between rats and humans compared to mice, higher breeding costs, greater difficulty in genetic manipulation, and higher reagent expenses, an increasing number of researchers are shifting away from rat models in favor of mouse models4,10,11. Among these, the mouse SAH model established via endovascular perforation of the carotid artery is considered the most physiologically accurate. However, the small size of mice presents significant challenges, demanding advanced surgical expertise from researchers12.
Detailed protocols for vascular perforation in mice remain scarce, with key difficulties including strain-specific differences, fine and anatomically complex vasculature, limited visibility of intracranial segments during surgery, difficulty in controlling bleeding, and high mortality rates12. To address these challenges, we improved upon previously described surgical procedures, meticulously recorded and analyzed intraoperative parameters, and performed comprehensive neurological evaluations post-modeling. These modifications enabled us to establish a more stable and standardized mouse SAH model, reducing surgical complexity and improving postoperative survival rates.
Materials and methods
Materials
The general instruments in this experiment included ophthalmic forceps, ophthalmic scissors, micro spring scissors, micro-ophthalmic forceps, and an electrocautery pen. For ligation, 7 − 0 black silk sutures (103-S, Teleflex, USA) were used. For puncturing, non-absorbable 4 − 0 polypropylene sutures (W8557, Ethicon, USA) were applied11.
Animals and grouping
Male C57BL/6 mice (10–12 weeks, 25–30 g) were obtained from the Animal Experiment Center of Nantong University. All mice were housed in a specific pathogen-free (SPF) environment at 23–25 °C with 55–60% humidity and maintained on a standard diet under a 12:12 h light/dark cycle. The experiments were approved by the Animal Ethics Committee of Nantong University (Approval No. S20201113-903). The mice were randomly divided into two groups: the sham group, where the filament touched the intracranial vascular wall without penetration, and the SAH group, where vascular puncture was performed to induce SAH. The temporal design of the entire investigation was illustrated in Supplementary Figure S1 and Table S1.
Mice were familiarized with the experimenter by being handled for 1–2 min daily for 7 days. For the first 3 days, they were allowed to freely explore the open field box for 30 min to familiarize themselves with the environment. On the experiment day, mice were placed in the experimental area 1 h(h) before testing to adapt to the environment.
Modified surgical procedure for the SAH intraluminal perforation model
After isoflurane anesthesia, the mice were placed in a supine position with limbs fixed and the neck (Fig. 1a) elevated by 5–8 mm to improve visibility and align the cervical vessels (Fig. 1b). The external carotid artery (ECA) was isolated and ligated distally with a permanent knot, while the internal carotid artery (ICA) was isolated and secured with a loose knot. The common carotid artery (CCA) was clamped using an arterial clamp (Fig. 1c) to minimize ischemic duration from ligation.
Schematic Workflow of the Intraluminal Puncture SAH Model in Mice. (a) Construction process of the SAH model via intraluminal vascular puncture. (b) Diagram showing the names and anatomical distribution of relevant blood vessels. (c) Ligation of the ICA and ECA with fine sutures, with the RCA clamped. (d) Isolation of the ECA and creation of a small incision at its proximal end. (e) Insertion of the filament through the ECA incision and advancement into the ICA after reversal. (f) Filament advancement into the intracranial region, perforating the vascular wall. (g) Filament withdrawal and ECA ligation. Note: Dashed lines indicate the subsequent repositioning of vascular branches. The blue filament represents a 4 − 0 monofilament nylon suture. SAH, subarachnoid hemorrhage; RCA, right common carotid artery; ICA, internal carotid artery; OA, posterior auricular artery; ECA, external carotid artery; STA, superficial temporal artery.
The ECA was cauterized distal to the knot using an electrocautery pen, and the proximal end was trimmed to create an incision (Fig. 1d). A sharpened 18 mm filament was inserted through the incision along the ECA into the bifurcation of the CCA. A loose knot was tied around the filament on the ECA to secure it and prevent backflow (Fig. 1e) without adding resistance to the filament’s advancement.
After releasing the ICA knot and the CCA clamp to restore blood flow, the filament was advanced at a 20–25° angle relative to the trachea into the ICA. Upon advancing 5 mm past the bifurcation, resistance indicated the external opening of the carotid canal. Adjusting the angle to 15–20°, the filament was advanced 13 mm in total until a distinct elastic resistance was felt, marking the endpoint (Fig. 1f). The filament tip was positioned within the right-side vessels of the Circle of Willis. With slight force, the filament was pushed 1–2 mm further, producing a clear sense of membrane rupture. No further advancement was performed to avoid puncturing brain tissue, which could lead to complications such as hemiplegia.
To prevent excessive bleeding and reduce mortality, the CCA was temporarily clamped for approximately 10 s after the rupture11. The filament was then withdrawn, and the ECA loose knot was tightened to seal the vessel (Fig. 1g). Finally, the neck skin was sutured, and the mice were placed on a warming blanket until they regained consciousness. Their recovery status was monitored to determine the success of the modeling procedure. The experiment was repeated three times, and the success rate, complications (hemiplegic paralysis), and mortality of the modified model were observed at 3 h, 6 h, 12 h, 24 h, and 72 h post-SAH.
Filament advancement measurement and blood observations
During surgery, the length from the filament’s tail to the bifurcation of the CCA was measured when the 18 mm filament tip reached the soft, elastic intracranial vessels. This distance represented the length from the CCA bifurcation to the puncture site and was correlated with the mouse’s body weight. The vascular condition on the dorsal skull surface was assessed using laser speckle contrast imaging (LSCI). Due to the transparency of the skull, blood diffusion could be directly observed with the naked eye, allowing immediate visualization of the bleeding on the dorsal skull and brain surface post-surgery. SAH occurs immediately after modeling, allowing for cerebrospinal fluid (CSF) collection immediately post-procedure. The mouse is placed in a prone position with the occipital region elevated. The occipital cistern is surgically exposed through dissection of the occipital area, where blood accumulation in the cisterna magna can be observed. A 0.3 mL insulin syringe is used to puncture and aspirate CSF. Depending on the mouse’s body weight and the proficiency of the experimenter, 5–15 µL of CSF can be collected. And CSF was collected from the cisterna magna for microscopic examination. The success of the SAH model is determined by the presence of blood accumulation in the cisterna magna and blood dispersion over the cranial vault, supplemented by behavioral assessments. The final confirmation is based on the identification of hemorrhagic sites and blood clots upon cranial dissection at the time of euthanasia, serving as the gold standard. The final mortality rate of mice during the early brain injury phase (0–72 h) is recorded. Mice that survive this critical period rarely experience subsequent mortality. All remaining mice are euthanized at 7 days post-SAH. Meanwhile, mouse body weights were recorded daily from day 0 to day 7 post-surgery.
Gait refinement analysis
A detailed gait analysis was performed on sham and SAH mice 24 h before and 48 h after surgery using the Mouse Gait Analysis System (SA114, Scansci Biotechnology, Jiangsu, China). The system features a green-illuminated glass runway, with a cover equipped with red LED lights to create silhouettes of the mice running through the corridor. A high-speed camera positioned beneath the runway records scattered light from the paw prints, which is digitized and analyzed by the gait analysis software.
After calibrating the runway and setting the detection area, mice underwent at least 30 min of training daily for three days to acclimate to the system. During each test, mice walked freely on the glass runway at least three times. Paw prints were automatically classified as right forepaw, right hind paw, left forepaw, and left hind paw. The software then marked and recorded the prints, analyzing gait parameters, including stride cycle, step length, contact area, and interlimb coordination.
Modified garcia score
The modified Garcia scoring method was used to assess neurological function one day before surgery and on the second day post-surgery. Neurological deficits were evaluated with an overall score ranging from 0 to 18, based on six tests: spontaneous activity (5 min in the cage), spontaneous limb movement, forelimb extension (while holding the tail), wire grid climbing, response to trunk-side touches, and response to whisker touches. Higher scores indicate better neurological function.
Open field test (OFT)
The open field test, analyzed using ANY-maze software (Stoelting, USA), was used to evaluate general motor abilities. The open field apparatus, made of white opaque acrylic, was used for habituation by allowing mice to explore freely for 30 min daily for three days prior to testing to minimize interference.
During the test, a camera was activated, and the mouse was placed in the open field apparatus (Stoelting, USA) to move and explore freely for 5 min. After each trial, the apparatus was thoroughly cleaned with alcohol to eliminate odors. The total distance traveled in 5 min was recorded as the baseline one day before surgery and measured again 48 h post-surgery for analysis.
LSCI assay
To assess cerebral blood flow changes following vascular perforation, we used an LSCI system (RWD Life Science Co., Ltd., Shenzhen, China). Ten minutes after deep anesthesia and modeling, the mouse’s head was fixed in a stereotaxic frame, and erythromycin eye ointment was applied to prevent corneal dryness. The skull was exposed through an incision in the scalp, with saline applied to keep it moist. LSCI was performed at various time points (1 h, 24 h, 48 h, and 72 h) before and after SAH induction. At each time point, the mice are anesthetized and cerebral blood flow was monitored continuously for 10 min, with data from the 2nd to 9 th minutes used for statistical analysis. Three major vessels in each hemisphere (left and right), within 2 mm diameter circles, were selected as regions of interest (ROI), and blood flow was measured in these ROIs.
Tissue preparation
Two days post-modeling, mice were deeply anesthetized with isoflurane and perfused with saline, followed by 4% paraformaldehyde. The brains were dissected and fixed overnight in 4% paraformaldehyde at 4 °C. They were then soaked in 20% sucrose in 0.1 M phosphate buffer for one day, followed by 30% sucrose for another day. Once the brains sank to the bottom of the 30% sucrose solution, they were sectioned into 15-µm coronal frozen sections using a cryostat (Leica CM1950). For sham and experimental mice, 1 h and 24 h post-surgery, PBS perfusion was performed through the heart, and blood diffusion at the skull base was examined.
Luxol fast blue (LFB) staining
LFB staining was used to visualize the morphology and pathological changes of neural myelin sheaths. Brain sections were washed with PBS, mounted on slides, and baked in an oven for 30 min to prevent detachment. The sections were incubated in pre-warmed LFB staining solution (MasterTech, USA) at 60 °C for 6–8 h, followed by rapid differentiation in 0.05% lithium carbonate solution. After differentiation, the sections were sequentially dehydrated in 75%, 95%, and 100% ethanol, and immersed in fresh xylene for 5 min. The sections were sealed with neutral resin and imaged using a brightfield microscope (DM4000B, Leica, Germany). Fluorescence intensity of corpus callosum and striatum was quantified using ImageJ software.
Immunofluorescence staining
Brain sections were reheated at room temperature for 30 min and permeabilized with 0.3% Triton X-100 for 10 min. After washing with PBS three times (10 min each), the sections were blocked with 10% donkey serum for 1 h at room temperature. They were then incubated overnight at 4 °C with primary antibodies Anti-NeuN (1:3000, Abcam, USA), Anti-myelin basic protein (MBP) (1:3000, Abcam, USA), Anti-neurofilament H (NF-H, SMI-32) (1:3000, BioLegend, USA), Anti-NG2 (1:3000, Santa Cruz, Bolivia), Anti-Olig2 (1:3000, Millipore, USA). The appropriate secondary antibodies were applied and incubated for 2 h at room temperature for visualization, Alexa Fluor® 488 AffiniPure Donkey Anti-Rabbit IgG (1:800, Jackson, USA), Alexa Fluor® 488 AffiniPure Donkey Anti-Mouse IgG (1:800, Jackson, USA), Cy™3 AffiniPure Donkey Anti-Mouse IgG (1:800, Jackson, USA), Cy™3 AffiniPure Donkey Anti-Rabbit IgG (1:800, Jackson, USA), Alexa Fluor® 647-conjugated AffiniPure Donkey Anti-Goat IgG (1:800, Jackson, USA). The stained sections were visualized and imaged using a confocal microscope (TCS SP8, Leica, Germany). Fluorescence intensity of corpus callosum was quantified using ImageJ software.
Statistical analysis
The data was performed using GraphPad Prism and Origin software. Baseline measurements were taken for all subjects prior to modeling, and post-modeling data were normalized by dividing by the corresponding baseline values to eliminate individual variability and ensure comparability. For data following a normal distribution, an independent sample t-test was used to compare differences between groups. For non-normally distributed data, non-parametric tests (Mann-Whitney U test or Kruskal-Wallis test) were applied. Statistical significance was defined as *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. All significance tests were two-tailed, and results are presented as mean ± standard error (Mean ± SEM).
Results
4 − 0 monofilament nylon: the ideal material for intraluminal vascular puncture
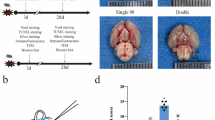
The choice and preparation of materials are crucial for surgical success and minimizing blood loss. Nylon filaments offer excellent hydrophobic and smooth surface properties. Through direct comparison of various nylon filament diameters with the carotid artery diameter in 10–12-week-old mice (Fig. 2a-b), we identified 3 − 0, 4 − 0, and 5 − 0 filaments as appropriate candidates. Further bending tests were conducted to assess the stiffness and flexibility of the three filaments (Fig. 2c-d). The 4 − 0 filament demonstrated superior flexibility over the 3 − 0 filament, facilitating passage through vascular curves. Additionally, it provided better structural support than the 5 − 0 filament, preventing bending during advancement and perforation of the vessel wall. To enhance success rates and control bleeding, we refined the filament tips. Trimming burrs and maintaining a sharp angle of 30–45 degrees (Fig. 2e-f) optimized the tactile sensation of vessel penetration and reduced bleeding. Based on these findings, the 4 − 0 monofilament nylon, with a smooth, sharp 30–45-degree tip, proved to be the most suitable material for model establishment in 10–12-week mice.
Instruments Required for Mouse Vascular Puncture SAH Model, Comparison of Carotid Artery and Filament Diameters, and Filament Material Handling. (a) General instruments: electrocautery pen, retractor, two curved micro-ophthalmic forceps (14 cm, curved tip, 0.15 mm tip width), ophthalmic forceps (10 cm, curved), needle holder (14 cm), ophthalmic scissors (10 cm, straight tip), micro hemostatic clamp (round tip, straight, 18 cm long), and micro spring scissors (8 cm, straight). (b) The diameter of 4 − 0 monofilament nylon suture (0.199–0.249 mm) is approximately 1/3 to 1/2 of the carotid artery diameter in 10–12-week-old mice. (c) Comparison of diameters for 3 − 0, 4 − 0, and 5 − 0 monofilament nylon sutures, each 18 mm in length, shown from left to right. (d) Comparison of the recovery of 3 − 0, 4 − 0, and 5 − 0 monofilament nylon suture materials after being folded under the same force, angle, and duration. (e, f) The tip of the filament material is modified to form a smooth, sharp angle of 30–45 degrees. Note: SAH, subarachnoid hemorrhage.
Blood accumulation and distribution after intraluminal perforation
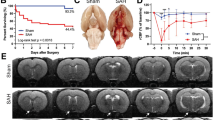
Within 1 h after intraluminal perforation, anatomical examination of the cisterna magna revealed distinct differences between the sham and SAH groups. In the sham group, the CSF was clear and colorless (Fig. 3a). In the SAH group, the cisterna magna was filled with red blood, with granular red blood cell deposits visible (Fig. 3b). Observation of the skull base showed no blood residue in the sham group. In the SAH group, the skull base was covered with blood within 1 h, with the highest accumulation at the puncture site and the brainstem, where clots had started to form. By 24 h, the blood at the skull base had significantly decreased, though small residual clots remained, primarily at the puncture site and brainstem, with slight variations elsewhere (Fig. 3c). Sagittal sections of brain tissue in the SAH group, taken 1 h post-modeling, revealed blood infiltration into brain spaces, extending to the hippocampus and white matter regions (Fig. 3d).
Observations of the Cisterna Magna and Cerebrospinal Fluid 1 h Post-Operation, and Blood Diffusion in the Skull Base and Sagittal Section. (a, b) Images of the cisterna magna and CSF 1 h post-operation in the sham and SAH groups. (c) Blood diffusion at the skull base in sham and SAH groups at 1 and 24 h post-operation (scale bar = 5 mm). (d) Sagittal brain tissue sections in sham and SAH groups 1 h post-modeling (scale bar = 2 mm). Note: SAH, subarachnoid hemorrhage; CSF, cerebrospinal fluid.
Modified model reduces mortality, standardizes advancement depth, and monitors weight changes
Statistical analysis of three repeated experiments with the modified model showed a 72-hour overall success rate of 91.43%. The primary complication was hemiplegia, characterized by severe contralateral paralysis with complete loss of strength, accompanied by rolling movements. Other cases involved contralateral motor dysfunction resulting from the pathophysiological effects of SAH, which could gradually recover over time. The incidence was 9.36%, and the mortality rate was 6.25% (Fig. 4a). Model failures were primarily due to difficulties in intracranial advancement of the filament, which post-mortem analysis revealed was caused by the complex and curved pathway of the internal carotid artery as it enters the skull. To enhance precision and standardization of the model, we measured the distance from the bifurcation of the CCA to the puncture site, finding the optimal average distance to be 13 ± 1 mm with no significant correlation to mouse body weight (Fig. 4b). Post-surgery, mice experienced a rapid weight decline, reaching a minimum within 72 h, followed by gradual recovery to normal levels (Fig. 4c). Mice with sustained weight loss that did not recover did not survive. Notably, higher-weight mice exhibited better tolerance and lower mortality, possibly due to greater energy reserves.
Relevant Parameters of the Modified SAH Model Construction. (a) The overall success rate, complication rate, and mortality rate of the modified model in 3, 6, 12, 24, 72 h across three repeated experiments under identical conditions. (b) Relationship between the distance from the carotid bifurcation to the puncture site and mouse body weight, with a mean distance of 13 ± 1 mm (n = 30). (c) Daily body weight changes in mice within 7 days post-modeling. Body weight gradually decreases, reaching the lowest point on day 3, followed by recovery to normal levels (n = 9). Note: SAH, subarachnoid hemorrhage.
Assessment of neurological impairment after SAH modeling
Gait analysis revealed more disorganized movement patterns in the SAH group compared to the sham group (Fig. 5a-b). The stride cycle of all four limbs was significantly shorter in the SAH group, with poorer contralateral coordination in the right forelimb and a decreasing trend in left forelimb function. However, there were no significant differences in hind limb coordination between the two groups. The stride length of the left forelimb and hind limb was significantly shorter in the SAH group compared to the sham group, with no notable difference in the right limbs. Additionally, the SAH group showed a larger peak contact area for both hind limbs, while no significant difference was observed in the forelimbs (Fig. 5c). In the modified Garcia scoring system, the neurological scores of the SAH group were significantly lower than those of the sham group, indicating impaired neurological function (Fig. 5d). In the OFT, the behavior of SAH mice was more disorganized compared to the sham group, with a significantly shorter walking distance within 5 min (Fig. 5e-f).
Behavioral Changes in Mice 48 h After Modified SAH Modeling. (a) Gait footprint analysis in sham and SAH groups, showing RF, LF, RH, and LH. (b) Paw pressure sequence diagram during gait analysis within the same time frame in sham and SAH groups, with RF (yellow), LF (red), RH (blue), and LH (green). (c) Statistical analysis of gait parameters, including stride cycle, step length, contact area, and interlimb coordination (n = 6). (d) Modified Garcia score comparison between sham and SAH groups (n = 6–8). (e, f) Total walking distance and corresponding trajectory plots and heatmaps from the open field test in sham and SAH groups. (n = 7). Note: *P < 0.05, ** P < 0.01, *** P < 0.001, **** P < 0.001 vs. sham group. SAH, subarachnoid hemorrhage, RF, right forepaw; LF, left forepaw; RH, right hind paw; LH, left hind paw.
Progressive reduction and recovery of cerebral blood flow after intraluminal perforation
Following modeling, varying degrees of blood overflow were observed on the brain surface, spreading from the anterior fontanelle downward and outward (Fig. 6a), marking a key indicator of successful SAH induction. To investigate CBF changes post-modeling, we used LSCI to measure CBF in the left and right hemispheres at 1 h pre-surgery and 1 h, 24 h, 48 h, and 72 h post-surgery (Fig. 6b-c). Before surgery, the cerebral blood flow was stable and evenly distributed across both hemispheres. One hour after the perforation procedure, a sharp decrease in CBF was observed, with a more pronounced reduction in the right hemisphere (the side of the puncture) than left hemisphere. Over the next 24 h and 48 h, blood flow in both hemispheres gradually recovered, with interhemispheric differences diminishing. By 72 h, CBF levels approached the pre-surgery baseline.
Changes in Cerebral Blood Flow in Mice at 1 h, 24 h, 48 h, and 72 h Pre- and Post-SAH. (a) Representative dorsal skull images from left to right, showing sham, mild, moderate, and severe hemorrhage. (b) Changes in cerebral blood flow in the left and right hemispheres of the brain at 1 h pre-operation, and at 1 h, 24 h, 48 h, and 72 h post-operation. Results are expressed as a percentage of the initial cerebral blood flow measured 1 h before SAH induction. (c) Representative heatmaps of cerebral blood flow captured by LSCI of the dorsal skull vasculature in mice at 1 h pre-operation, and at 1 h, 24 h, 48 h, and 72 h post-operation. Note: SAH, subarachnoid hemorrhage, LCH, left cerebral blood flow hemispheres; RCH, right cerebral blood flow hemispheres; LSCI, laser speckle contrast imaging.
White matter injury in the corpus callosum and striatum after SAH in mice
LFB staining showed uniform and deep blue coloration in white matter-rich regions, such as the corpus callosum and striatum (bregma − 0.50, −0.34 mm), indicating dense myelinated nerve fibers in sham mice. In contrast, the same regions in the SAH group exhibited lighter and uneven staining with fragmented patterns (Fig. 7a-b). Quantitative analysis confirmed a significant reduction in LFB staining intensity in the SAH group compared to the sham group (Fig. 7c), suggesting white matter damage after SAH modeling. Immunofluorescence staining with MBP (a marker of myelin) and SMI-32 (a marker of neurofilament degradation) in the corpus callosum. An increased SMI-32/MBP ratio in the SAH group compared to the sham group indicating damage to myelinated fibers following intraluminal perforation, highlighting the impact of SAH on white matter integrity (Fig. 7d-e).
White Matter myelin Injury in the corpus callosum and Striatum After SAH in mice. (a) Luxol fast blue staining of the corpus callosum and striatum in coronal brain sections (bregma − 0.5 mm, −0.34 mm) in the sham and SAH groups. Scale bar = 500 μm. (b) Enlarged views of Luxol fast blue-stained corpus callosum and striatum in coronal sections (bregma − 0.5 mm, −0.34 mm) from the sham and SAH groups. Scale bar = 200 μm. (c) Quantitative analysis of Luxol fast blue staining intensity in the corpus callosum and striatum of coronal sections (bregma − 0.5 mm, −0.34 mm) in the sham and SAH groups. Data are presented as the staining intensity (n = 3–4). (d) Representative images of MBP and SMI-32 immunofluorescence in the corpus callosum of sham and SAH mice, shown in coronal sections. Scale bar = 100 μm. (e) Quantitative fluorescence analysis of SMI-32/MBP intensity ratio in the corpus callosum of the sham and SAH groups, n = 3. Note: *P < 0.05, ** P < 0.01 vs. sham group. SAH, subarachnoid hemorrhage; MBP, Myelin Basic Protein; SMI-32, A marker for neurofilament protein; DAPI: 4′,6-diamidino-2-phenylindole (a nuclear stain).
Oligodendrocyte injury and proliferation, and neuronal damage in the cortex and striatum after SAH in mice
In the corpus callosum, significant differences in Olig2 and NG2 expression were observed between the sham and SAH groups (Fig. 8a). In the sham group, Olig2-positive oligodendrocytes were abundant and evenly distributed. However, in the SAH group, the number of Olig2-positive cells decreased significantly, with reduced fluorescence intensity (Fig. 8b), indicating oligodendrocyte injury and myelin loss. NG2-positive cells (oligodendrocyte precursor cells, OPCs) were minimally expressed in the sham group but markedly increased in the SAH group, with enhanced fluorescence intensity (Fig. 8c), suggesting active OPC proliferation involved in tissue repair. The co-expression analysis of Olig2 and NG2 revealed that post-SAH, OPCs not only proliferated but also differentiated, contributing to remyelination. In the cortex and striatum, NeuN-labeled neurons showed significant differences between the sham and SAH groups (Fig. 8d-g). In the sham group, neurons in both the cortex and striatum exhibited high fluorescence intensity, indicating a widespread distribution of normal neurons. In contrast, the SAH group displayed a marked reduction in NeuN fluorescence intensity (Fig. 8d-g), signifying extensive neuronal damage and death in these regions post-SAH.
Oligodendrocyte Injury and Proliferation, and Neuronal Damage in the Cortex and Striatum After SAH in mice. (a) Immunofluorescence images of Olig2 and NG2 in the corpus callosum (bregma − 0.5 mm) of sham and SAH mice. Scale bar = 100 μm. (b-c) Quantitative analysis of Olig2 and NG2 fluorescence in the corpus callosum of sham and SAH mice. n = 3. (d-g) Immunofluorescence images of NeuN staining in the cortex and striatum of sham and SAH mice. Scale bar = 100 μm. Quantitative analysis of NeuN fluorescence in the cortical and striatal regions of sham and SAH mice. n = 3. Note: * P < 0.05, ** P < 0.01, vs. sham group. SAH, Subarachnoid Hemorrhage; Olig2, Oligodendrocyte Transcription Factor 2; NG2: Neural/Glial Antigen 2 (marker for oligodendrocyte precursor cells); DAPI: 4′,6-diamidino-2-phenylindole (a nuclear stain); NeuN: Neuronal Nuclei (a marker for neurons).
Discussion
We have reviewed and analyzed previous methods for constructing in vivo SAH models and have provided detailed improvements and descriptions of the surgical techniques and materials used. To evaluate how well this model simulates the pathophysiological progression of SAH, we conducted anatomical observations, behavioral assessments, post-modeling monitoring of cerebral blood flow, and, finally, histological evaluation of myelin and neuronal damage in the white matter regions of the brain. Compared with rats, using mice is less costly and requires fewer reagents. Besides, their smaller cranial cavity makes the intracranial high-pressure environment more intense13,14, which is closer to the acute changes after human SAH. Moreover, their higher gene homology also makes their pathophysiological changes more similar to those in humans14,15,17. Through these optimizations, we successfully simulated the pathophysiological processes of human SAH in a mouse model.
Although numerous studies have used mouse intraluminal perforation models9,18,19, there is still no standardized surgical protocol due to the technical challenges and complexity of mouse anatomy. Specific details and parameters during the procedure are rarely described. To address this, we conducted experiments on mice and found that using 12-week-old mice and a 4 − 0 filament, with an insertion depth of 13 mm from the carotid bifurcation, greatly improved the success rate to over 95%. Ligation of the external carotid artery after guidewire insertion may transiently increase ipsilateral ICA blood flow. However, studies demonstrate compensatory mechanisms: bilateral carotid flow redistribution, intracranial hemodynamic balance via the Circle of Willis, and metabolic-linked vascular regulation19,20,21,23. These mechanisms work together through multiple pathways to maintain cerebral blood flow stability across stages. Our study identified the age of the mice and the selection and modification of filament material as key factors influencing success and stability. The consistent bleeding point between the middle cerebral artery and anterior cerebral artery corroborates previous findings12.
To standardize this model, we first conducted anatomical observations of the brains of mice post-SAH modeling. Due to the differences in body size and the sustained nature of the hemorrhage, mouse hemorrhage differs from that of rats, and the rat grading scale for hemorrhage is not applicable to mice24. Observing the cisterna magna, we found that within 1 h, the CSF changed from clear to opaque red, reflecting the pathological process of SAH. Anatomical examination 1 h and 48 h post-SAH revealed that blood spread outward from the puncture site within the first hour, aligning with previous studies25. However, by 48 h, the amount of blood in the mouse brain had significantly decreased, primarily remaining near the puncture site and brainstem. This pattern of blood distribution may be related to post-SAH neurological sequelae such as meningeal irritation, hydrocephalus, dizziness, vomiting, and cognitive impairment observed in clinical patients26,27. Sagittal sections showed blood diffusion across the brain surface, including the white matter regions1,28,29,30,31, demonstrating the damaging effects of SAH on white matter. This likely contributes to the motor deficits observed in many clinical SAH patients2,32. These stable pathological manifestations offer researchers a reliable platform for drug testing and the development of SAH treatments33,34,35.
There is growing interest among researchers in early brain injury and white matter damage, which leads to cognitive and motor impairments after SAH36. Considering the precision required for behavioral experiments, a highly stable and reproducible animal model is essential. Behavioral experiments often yield unsatisfactory results due to technical variability5, complicating their implementation and limiting their application. Our study utilized modified Garcia scores and open field tests 48 h post-SAH, revealing significant motor impairment in the SAH group. These results were consistent with both other animal models and clinical observations37,38,39,40. Unlike studies of other neurological diseases like Parkinson’s disease41,42, research on gait behavior following SAH is limited, with few studies on early brain injury. Our improved model’s enhanced stability and reproducibility allowed for a detailed analysis of gait in post-SAH mice. We observed specific impairments in contralateral forelimb coordination, hindlimb support capacity, and walking ability, as well as overall stride cycle abnormalities, which mirror neurological deficits seen in clinical patients43. These findings may guide future research on therapeutic targets for SAH-related motor impairments44.
Vasospasm remains a key topic in SAH research, with changes in cerebral blood flow being a critical indicator of model reliability45. In our model, we observed direct blood diffusion across the skull surface, which was not associated with mortality46. ICP plays a significant role in SAH. The most immediate event following SAH is a sudden increase in ICP and cessation of intracranial circulation, leading to a sharp decline in CBF, which is the first step in the pathological cascade that results in cytotoxic edema formation, endothelial cell apoptosis, and blood-brain barrier (BBB) disruption47. The elevation of ICP leads to a reduction in cerebral perfusion pressure (CPP), thereby initiating initial ischemic injury. Compared to rats, the cranial cavity of mice is more confined, and within 72 h after SAH, they experience higher ICP and lower CBF48,49, which is consistent with the results of our laser speckle flowmetry experiments on CBF. During the early brain injury (EBI) period of SAH, with the increase of ICP and arterial blood stimulation, acute vasospasm occurs, and CBF drops sharply, further inducing global ischemic brain injury. Given the acute disturbances in cerebral blood flow observed after SAH, it is essential to distinguish global ischemia associated with EBI from focal ischemia resulting from inadvertent middle cerebral artery (MCA) occlusion. To achieve this, we performed anatomical, pathological, and functional evaluations to verify the integrity of the MCA and confirm the global nature of the ischemic injury. Anatomically, the MCA in mice is a smaller branch arising from the Circle of Willis, and the filament used for SAH induction is too large in diameter to enter or occlude the MCA. Postmortem examination (Fig. 3C) revealed blood clot accumulation predominantly around the Circle of Willis, with intact MCA vessels and no evidence of rupture or occlusion. Furthermore, laser speckle contrast imaging demonstrated a relatively uniform reduction in cerebral blood flow across the hemisphere, rather than a focal perfusion defect confined to the MCA territory. Together, these findings confirm that hemispheric ischemia due to MCA occlusion is unlikely in our model.
Due to the repair of the bleeding point and the acute compensatory mechanism of intracranial blood flow, the impact of acute vasospasm on prognosis is not obvious. With the passage of time, the harm caused by the pathological and physiological cascade reactions such as neuroinflammation, blood-brain barrier damage and smooth muscle dysfunction caused by ischemic brain injury begins to amplify, and delayed cerebral vasospasm occurs, which has an important impact on the prognosis of patients50,51. Compared to costly or invasive techniques like MRI or intracranial pressure monitoring52, direct observation offers a low-cost, minimally invasive method to assess model success, improving the accuracy of subsequent studies. Early brain injury has been shown to involve acute vasospasm, which plays an important role in cortical spread and neurovascular coupling53,54, significantly affecting prognosis55,56. Using laser speckle contrast imaging, we observed an acute reduction in cerebral blood flow and blanching of the hemisphere following perforation, consistent with acute vasospasm post-SAH57. The combination of hemorrhage and ischemia contributes to the poor outcomes associated with SAH.
Clinically, the neurological and motor functions of SAH patients are a primary concern. Nearly all SAH patients experience white matter involvement58,59, making white matter damage a critical point in early brain injury28. In our model, Luxol fast blue staining revealed significant white matter damage in the corpus callosum and striatum within 48 h, aligning with our behavioral results. Immunofluorescence staining showed a shift from mature to immature myelin post-SAH, reflecting white matter injury and repair28. We also observed oligodendrocyte damage and active proliferation in the corpus callosum post-SAH, providing a more representative pathological feature compared to rat or single-injection models29. This highlights the model’s utility for further therapeutic research60. Additionally, we found severe and sustained neuronal damage in various brain regions, offering a physiologically relevant platform to test therapeutic strategies.
Despite its advantages, our study has limitations. It has been reported that mouse strain and sex may influence the pathophysiological changes of SAH, possibly due to differences in estrogen and androgen levels61,62,63,64. Furthermore, weaker or malnourished mice may have higher mortality rates, and anatomical differences between strains may affect the perforation site12. While early brain injury is increasingly recognized as a key factor in SAH, long-term disease progression also requires further investigation.
In conclusion, our study provides an improved mouse SAH model and optimized evaluation methods, providing a new tool and more effective technique for studying the related injuries during the early brain injury period of subarachnoid hemorrhage. This innovative model not only better simulates and assesses the pathological processes of SAH but also lays a solid foundation for future drug screening and therapeutic research.
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Pang, J. et al. White matter injury in early brain injury after subarachnoid hemorrhage. Cell Transplant. 28, 26–35. https://doi.org/10.1177/0963689718812054 (2019).
Lv, B. et al. Epidemiological trends of subarachnoid hemorrhage at global, regional, and National level: a trend analysis study from 1990 to 2021. Military Med. Res. 11, 46. https://doi.org/10.1186/s40779-024-00551-6 (2024).
Bühler, D., Schüller, K. & Plesnila, N. Protocol for the induction of subarachnoid hemorrhage in mice by perforation of the circle of Willis with an endovascular filament. Translational Stroke Res. 5, 653–659. https://doi.org/10.1007/s12975-014-0366-6 (2014).
Liu, S. et al. Endovascular perforation model for subarachnoid hemorrhage combined with magnetic resonance imaging (MRI). J. Visualized Experiments: JoVE. https://doi.org/10.3791/63150 (2021).
Alpdogan, S., Li, K., Sander, T., Cornelius, J. F. & Muhammad, S. Cisterna magna injection mouse model of subarachnoid hemorrhage (SAH): A systematic literature review of preclinical SAH research. J. Experimental Neurol. 4, 11–20. https://doi.org/10.33696/Neurol.4.069 (2023).
Lai, J. H., Qin, T., Sakadžić, S., Ayata, C. & Chung, D. Y. Cortical spreading depolarizations in a mouse model of subarachnoid hemorrhage. Neurocrit. Care. 37, 123–132. https://doi.org/10.1007/s12028-021-01397-9 (2022).
Broderick, J. P., Brott, T. G., Duldner, J. E., Tomsick, T. & Leach, A. Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke 25, 1342–1347. https://doi.org/10.1161/01.str.25.7.1342 (1994).
Feiler, S., Friedrich, B., Schöller, K., Thal, S. C. & Plesnila, N. Standardized induction of subarachnoid hemorrhage in mice by intracranial pressure monitoring. J. Neurosci. Methods. 190, 164–170. https://doi.org/10.1016/j.jneumeth.2010.05.005 (2010).
Puglisi, C. H. et al. Sustained ICP elevation is a driver of Spatial memory deficits after intraventricular hemorrhage and leads to activation of distinct microglial signaling pathways. Translational Stroke Res. 14, 572–588. https://doi.org/10.1007/s12975-022-01061-0 (2023).
Mutoh, T. et al. Inotropic support against early brain injury improves cerebral hypoperfusion and outcomes in a murine model of subarachnoid hemorrhage. Brain Res. Bull. 130, 18–26. https://doi.org/10.1016/j.brainresbull.2016.12.009 (2017).
Muroi, C. et al. Filament perforation model for mouse subarachnoid hemorrhage: surgical-technical considerations. Br. J. Neurosurg. 28, 722–732. https://doi.org/10.3109/02688697.2014.918579 (2014).
Weyer, V. et al. Influence of neurovascular anatomy on perforation site in different mouse strains using the filament perforation model for induction of subarachnoid hemorrhage. PloS One. 17, e0263983. https://doi.org/10.1371/journal.pone.0263983 (2022).
Dienel, A. et al. Confirming subarachnoid hemorrhage induction in the endovascular puncture mouse model. Brain Hemorrhages. 3, 111–116. https://doi.org/10.1016/j.hest.2022.02.001 (2022).
Barth, K. N., Onesti, S. T., Krauss, W. E. & Solomon, R. A. A simple and reliable technique to monitor intracranial pressure in the rat: technical note. Neurosurgery 30, 138–140. https://doi.org/10.1227/00006123-199201000-00028 (1992).
O’Brien, S. J. et al. Anchored reference loci for comparative genome mapping in mammals. Nat. Genet. 3, 103–112. https://doi.org/10.1038/ng0293-103 (1993).
Nadeau, J. H. Maps of linkage and synteny homologies between mouse and man. Trends Genet. 5, 82–86. https://doi.org/10.1016/0168-9525(89)90031-0 (1989).
Serikawa, T. et al. A comparative genetic map of rat, mouse and human genomes. Exp. Anim. 47, 1–9. https://doi.org/10.1538/expanim.47.1 (1998).
Kawakita, F., Nakano, F., Kanamaru, H., Asada, R. & Suzuki, H. Anti-Apoptotic effects of AMPA receptor antagonist perampanel in early brain injury after subarachnoid hemorrhage in mice. Translational Stroke Res. 15, 462–475. https://doi.org/10.1007/s12975-023-01138-4 (2024).
Bandyopadhyay, S. et al. Red blood cells in the cerebrospinal fluid compartment after subarachnoid haemorrhage: significance and emerging therapeutic strategies. Translational Stroke Res. https://doi.org/10.1007/s12975-024-01238-9 (2024).
Korshunov, V. A. & Berk, B. C. Flow-induced vascular remodeling in the mouse: a model for carotid intima-media thickening. Arterioscler. Thromb. Vasc Biol. 23, 2185–2191. https://doi.org/10.1161/01.Atv.0000103120.06092.14 (2003).
Kennedy McConnell, F. & Payne, S. The dual role of cerebral autoregulation and collateral flow in the circle of Willis after major vessel occlusion. IEEE Trans. Biomed. Eng. 64, 1793–1802. https://doi.org/10.1109/tbme.2016.2623710 (2017).
van Kooij, B. J., Hendrikse, J., Benders, M. J., de Vries, L. S. & Groenendaal, F. Anatomy of the circle of Willis and blood flow in the brain-feeding vasculature in prematurely born infants. Neonatology 97, 235–241. https://doi.org/10.1159/000253754 (2010).
Vriens, E. M. et al. Flow redistribution in the major cerebral arteries after carotid endarterectomy: a study with transcranial doppler scan. J. Vasc Surg. 33, 139–147. https://doi.org/10.1067/mva.2001.109768 (2001).
Sugawara, T., Ayer, R., Jadhav, V. & Zhang, J. H. A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J. Neurosci. Methods. 167, 327–334. https://doi.org/10.1016/j.jneumeth.2007.08.004 (2008).
Zhang, J. H. Vascular neural network in subarachnoid hemorrhage. Translational Stroke Res. 5, 423–428. https://doi.org/10.1007/s12975-014-0355-9 (2014).
Sehba, F. A., Hou, J., Pluta, R. M. & Zhang, J. H. The importance of early brain injury after subarachnoid hemorrhage. Prog. Neurobiol. 97, 14–37. https://doi.org/10.1016/j.pneurobio.2012.02.003 (2012).
Wang, M. D. et al. The role of early cerebral edema and hematoma assessment in aneurysmal subarachnoid hemorrhage (a-SAH) in predicting early brain injury (EBI) and cognitive impairment: a case controlled study. Int. J. Surg. (London England). 110, 3166–3177. https://doi.org/10.1097/JS9.0000000000001244 (2024).
Chen, M. et al. Myelin sheath injury and repairment after subarachnoid hemorrhage. Front. Pharmacol. 14 https://doi.org/10.3389/fphar.2023.1145605 (2023).
Peng, K. et al. A timeline of oligodendrocyte death and proliferation following experimental subarachnoid hemorrhage. CNS Neurosci. Ther. 28, 842–850. https://doi.org/10.1111/cns.13812 (2022).
Peng, J. et al. LRP1 activation attenuates white matter injury by modulating microglial polarization through Shc1/PI3K/Akt pathway after subarachnoid hemorrhage in rats. Redox Biol. 21, 101121. https://doi.org/10.1016/j.redox.2019.101121 (2019).
Fern, R. F., Matute, C. & Stys, P. K. White matter injury: ischemic and nonischemic. Glia 62, 1780–1789. https://doi.org/10.1002/glia.22722 (2014).
Rehman, S. et al. Case-Fatality and functional outcome after subarachnoid hemorrhage (SAH) in international stroke oUtComes sTudy (INSTRUCT). J. Stroke Cerebrovasc. Diseases: Official J. Natl. Stroke Association. 31, 106201. https://doi.org/10.1016/j.jstrokecerebrovasdis.2021.106201 (2022).
Yamanaka, T. et al. Preventive effect of intermittent cerebrospinal fluid drainage for secondary chronic hydrocephalus after aneurysmal subarachnoid hemorrhage. Fluids Barriers CNS. 20, 91. https://doi.org/10.1186/s12987-023-00486-5 (2023).
Zille, M., Plesnila, N. & Boltze, J. Pharmacologically targeting inflammation and improving cerebrospinal fluid circulation improves outcome after subarachnoid haemorrhage. EBioMedicine 77, 103937. https://doi.org/10.1016/j.ebiom.2022.103937 (2022).
Schallner, N. et al. Microglia regulate blood clearance in subarachnoid hemorrhage by Heme oxygenase-1. J. Clin. Investig. 125, 2609–2625. https://doi.org/10.1172/JCI78443 (2015).
Lauzier, D. C. et al. Early brain injury after subarachnoid hemorrhage: incidence and mechanisms. Stroke 54, 1426–1440. https://doi.org/10.1161/STROKEAHA.122.040072 (2023).
Al-Khindi, T., Macdonald, R. L. & Schweizer, T. A. Cognitive and functional outcome after aneurysmal subarachnoid hemorrhage. Stroke 41, e519–536. https://doi.org/10.1161/STROKEAHA.110.581975 (2010).
Chung, D. Y. et al. Subarachnoid hemorrhage leads to early and persistent functional connectivity and behavioral changes in mice. J. Cereb. Blood Flow. Metabolism: Official J. Int. Soc. Cereb. Blood Flow. Metabolism. 41, 975–985. https://doi.org/10.1177/0271678X20940152 (2021).
Boettinger, S. et al. Behavioral characterization of the anterior injection model of subarachnoid hemorrhage. Behav. Brain. Res. 323, 154–161. https://doi.org/10.1016/j.bbr.2017.02.004 (2017).
Boyko, M. et al. The neuro-behavioral profile in rats after subarachnoid hemorrhage. Brain Res. 1491, 109–116. https://doi.org/10.1016/j.brainres.2012.10.061 (2013).
Encarnacion, A. et al. Long-term behavioral assessment of function in an experimental model for ischemic stroke. J. Neurosci. Methods. 196, 247–257. https://doi.org/10.1016/j.jneumeth.2011.01.010 (2011).
Wang, X. H. et al. Quantitative assessment of gait and neurochemical correlation in a classical murine model of Parkinson’s disease. BMC Neurosci. 13, 142. https://doi.org/10.1186/1471-2202-13-142 (2012).
Yeo, S. S. et al. Evidence of corticospinal tract injury at midbrain in patients with subarachnoid hemorrhage. Stroke 43, 2239–2241. https://doi.org/10.1161/STROKEAHA.112.661116 (2012).
Zheng, Z. Y. et al. Integrated analysis of gait parameters and gene expression profiles in a murine model of subarachnoid hemorrhage. Genes Brain Behav. 20, e12728. https://doi.org/10.1111/gbb.12728 (2021).
Prunell, G. F., Mathiesen, T. & Svendgaard, N. A. Experimental subarachnoid hemorrhage: cerebral blood flow and brain metabolism during the acute phase in three different models in the rat. Neurosurgery 54, 426–436; discussion 436–437, (2004). https://doi.org/10.1227/01.neu.0000103670.09687.7a
Mutoh, T. et al. Simple procedure for assessing diffuse subarachnoid hemorrhage successfully created using filament perforation method in mice. Anim. Models Experimental Med. 7, 77–81. https://doi.org/10.1002/ame2.12372 (2024).
Solár, P., Zamani, A., Lakatosová, K. & Joukal, M. The blood-brain barrier and the neurovascular unit in subarachnoid hemorrhage: molecular events and potential treatments. Fluids Barriers CNS. 19, 29. https://doi.org/10.1186/s12987-022-00312-4 (2022).
Prunell, G. F., Mathiesen, T., Diemer, N. H. & Svendgaard, N. A. Experimental subarachnoid hemorrhage: subarachnoid blood volume, mortality rate, neuronal death, cerebral blood flow, and perfusion pressure in three different rat models. Neurosurgery 52, 165–175. https://doi.org/10.1097/00006123-200301000-00022 (2003). discussion 175 – 166.
Zoerle, T. et al. Intracranial pressure after subarachnoid hemorrhage. Crit. Care Med. 43, 168–176. https://doi.org/10.1097/ccm.0000000000000670 (2015).
Sabri, M., Lass, E. & Macdonald, R. L. Early brain injury: a common mechanism in subarachnoid hemorrhage and global cerebral ischemia. Stroke Res Treat 394036, (2013). https://doi.org/10.1155/2013/394036 (2013).
Topkoru, B., Egemen, E., Solaroglu, I. & Zhang, J. H. Early brain injury or vasospasm?? An overview of common mechanisms. Curr. Drug Targets. 18, 1424–1429. https://doi.org/10.2174/1389450117666160905112923 (2017).
Egashira, Y., Shishido, H., Hua, Y., Keep, R. F. & Xi, G. New grading system based on magnetic resonance imaging in a mouse model of subarachnoid hemorrhage. Stroke 46, 582–584. https://doi.org/10.1161/STROKEAHA.114.007834 (2015).
Tang, Y. et al. Cortical spreading depression aggravates early brain injury in a mouse model of subarachnoid hemorrhage. J. Biophotonics. 14, e202000379. https://doi.org/10.1002/jbio.202000379 (2021).
Em, H. Coupling mechanism and significance of the BOLD signal: a status report. Annu. Rev. Neurosci. 37 https://doi.org/10.1146/annurev-neuro-071013-014111 (2014).
Matsubara, H. et al. Involvement of cerebral blood flow on neurological and behavioral functions after subarachnoid hemorrhage in mice. J. Stroke Cerebrovasc. Diseases: Official J. Natl. Stroke Association. 30, 105952. https://doi.org/10.1016/j.jstrokecerebrovasdis.2021.105952 (2021).
Matsubara, H. et al. Importance of CBF measurement to exclude concomitant cerebral infarction in the murine endovascular perforation SAH model. J. Stroke Cerebrovasc. Diseases: Official J. Natl. Stroke Association. 29, 105243. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.105243 (2020).
Sun, B. L. et al. Dynamic alterations of cerebral Pial microcirculation during experimental subarachnoid hemorrhage. Cell. Mol. Neurobiol. 29, 235–241. https://doi.org/10.1007/s10571-008-9316-8 (2009).
Wang, Y. et al. White matter injury in ischemic stroke. Prog. Neurobiol. 141, 45–60. https://doi.org/10.1016/j.pneurobio.2016.04.005 (2016).
Reijmer, Y. D. et al. Microstructural White Matter Abnormalities and Cognitive Impairment After Aneurysmal Subarachnoid Hemorrhage. Stroke 49, 2040–2045, (2018). https://doi.org/10.1161/STROKEAHA.118.021622
Oluich, L. J. et al. Targeted ablation of oligodendrocytes induces axonal pathology independent of overt demyelination. J. Neuroscience: Official J. Soc. Neurosci. 32, 8317–8330. https://doi.org/10.1523/JNEUROSCI.1053-12.2012 (2012).
Koduri, S. et al. The two faces of Estrogen in experimental hemorrhagic stroke. Translational Stroke Res. 13, 362–363. https://doi.org/10.1007/s12975-021-00942-0 (2022).
Zhang, J. et al. Acute T2*-Weighted magnetic resonance imaging detectable cerebral thrombosis in a rat model of subarachnoid hemorrhage. Translational Stroke Res. 13, 188–196. https://doi.org/10.1007/s12975-021-00918-0 (2022).
Guo, Y. et al. Role of Estrogen-Related receptor γ and PGC-1α/SIRT3 pathway in early brain injury after subarachnoid hemorrhage. Neurotherapeutics: J. Am. Soc. Experimental Neurother. 20, 822–837. https://doi.org/10.1007/s13311-022-01330-8 (2023).
Islam, R. et al. Development of a 3D brain model to study Sex-Specific neuroinflammation after hemorrhagic stroke. Translational Stroke Res. https://doi.org/10.1007/s12975-024-01243-y (2024).
Funding
This research was supported by National Natural Science Foundation of China (Grant number: 82171190), Medical science and technology research (joint construction) project (Grant number: LHGJ20230032), Henan Center for Outstanding Overseas Scientists (Grant number GZS2022019), and Henan Province science and technology research project (Grant number: 242102311095).
Author information
Authors and Affiliations
Contributions
Q.L., J.G., and G.W. contributed to the conceptualization, supervision, project administration, and funding acquisition of the study. Q.L. and J.G. were involved in writing and reviewing the manuscript. C.X., F.L., and B.L. performed the experiments and collected data. W.G. and Z.Z. conducted data analysis and visualization, while H.W. and F.H. were responsible for data validation. Z.C. and L.S. inspected and validated the data. G.F. and L.Y. prepared the original draft and contributed to the manuscript review and editing. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Consent for publication
All authors have approved the manuscript and agree with its submission.
Competing interests
The authors declare no competing interests.
Ethical committee and informed consent
All animal experiments followed the guidelines set forth by the NIH Guide for the Care and Use of Laboratory Animals and were approved by the Animal Ethics Committee of Nantong University (Approval No. S20201113-903). The study is reported in accordance with ARRIVE guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xiang, C., Liu, F., Liu, B. et al. Optimization and evaluation of an experimental subarachnoid hemorrhage model in mice. Sci Rep 15, 18285 (2025). https://doi.org/10.1038/s41598-025-03016-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-025-03016-5